��Ŀ����
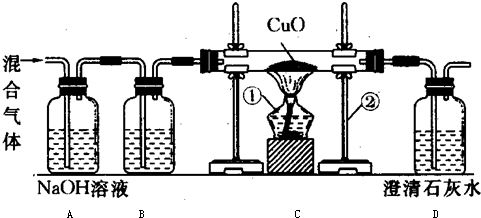
ij����С������һ����̼�ⶨCuO�Ļ�ԭ�ʣ�ʵ�鿪ʼʱ���Բ���ʵ�������δ���κδ�����ֱ��ʹ�ã������õ�CuO�Ļ�ԭ�ʳ�����l00%�����������������۲��Ľ�ʵ��װ�ã��Ľ����ʵ��װ�ü���õ�ʵ����������ͼ������
�Իش�
��1��ָ��ͼ������ʾ�����������ƣ���______����______��
��2���Ľ�ʵ��װ��ǰ��õ�CuO�Ļ�ԭ�ʳ�����100%������ܵ�ԭ��֮һ��ͨ���һ����̼�����л���______���壮
��3��Aװ���з�����Ӧ�Ļ�ѧ����ʽ��______
��4�������ɵ�������Dװ���б���ȫ���գ����õ�CuO�Ļ�ԭ��Ϊ______��
��5��Ϊ�˱���������Ӧ��Dװ�õ������ܿڲ�ȡ�Ĵ�ʩ��______��
| ������������g�� | 7 |
| CuO������g�� | 20 |
| ����������g�� | 20 |

�Իش�
��1��ָ��ͼ������ʾ�����������ƣ���______����______��
��2���Ľ�ʵ��װ��ǰ��õ�CuO�Ļ�ԭ�ʳ�����100%������ܵ�ԭ��֮һ��ͨ���һ����̼�����л���______���壮
��3��Aװ���з�����Ӧ�Ļ�ѧ����ʽ��______
��4�������ɵ�������Dװ���б���ȫ���գ����õ�CuO�Ļ�ԭ��Ϊ______��
��5��Ϊ�˱���������Ӧ��Dװ�õ������ܿڲ�ȡ�Ĵ�ʩ��______��
��1��ͼ������ʾ�����������ƣ��پƾ��ƣ�������̨��
��2��һ����̼��ԭ����ͭ���ɶ�����̼��Dװ���е�ʯ��ˮ���գ����Լ����CuO�Ļ�ԭ�ʣ��������������ж�����̼��Ҳ�ᱻD�е�ʯ��ˮ���գ���������CuO�Ļ�ԭ�ʾͻᳬ��100%��
��3����Aװ���У�������̼�����������Ʒ�Ӧ������̼���ƺ�ˮ����Ӧ�ķ���ʽ�ǣ�CO2+2NaOH=Na2CO3+H2O��
��4�������ɵĶ�����̼������Ϊx
CO2+Ca��OH��2�TCaCO3��+H2O
44 100
x 20g
=
x�T8.8g
�豻��ԭ������ͭ������Ϊy
CO+CuO
Cu+CO2
80 44
y 8.8g
=
y=16g
CuO�Ļ�ԭ��Ϊ��
��100%=80%��
��5�����������һ����̼�ж��������ų�װ���⣬����Ҫ����β��������Ӧ��Dװ�õ������ܿڲ�ȡ�Ĵ�ʩ�ǣ��������ܻ�Ϊ���쵼�ܲ���ȼ��
�ʴ�Ϊ����1���ƾ��ƣ�����̨�� ��2��������̼����3��CO2+2NaOH=Na2CO3+H2O����4��80%����5���������ܻ�Ϊ���쵼�ܲ���ȼ��
��2��һ����̼��ԭ����ͭ���ɶ�����̼��Dװ���е�ʯ��ˮ���գ����Լ����CuO�Ļ�ԭ�ʣ��������������ж�����̼��Ҳ�ᱻD�е�ʯ��ˮ���գ���������CuO�Ļ�ԭ�ʾͻᳬ��100%��
��3����Aװ���У�������̼�����������Ʒ�Ӧ������̼���ƺ�ˮ����Ӧ�ķ���ʽ�ǣ�CO2+2NaOH=Na2CO3+H2O��
��4�������ɵĶ�����̼������Ϊx
CO2+Ca��OH��2�TCaCO3��+H2O
44 100
x 20g
| 44 |
| 100 |
| x |
| 20g |
�豻��ԭ������ͭ������Ϊy
CO+CuO
| ||
80 44
y 8.8g
| 80 |
| 44 |
| y |
| 8.8g |
CuO�Ļ�ԭ��Ϊ��
| 16g |
| 20g |
��5�����������һ����̼�ж��������ų�װ���⣬����Ҫ����β��������Ӧ��Dװ�õ������ܿڲ�ȡ�Ĵ�ʩ�ǣ��������ܻ�Ϊ���쵼�ܲ���ȼ��
�ʴ�Ϊ����1���ƾ��ƣ�����̨�� ��2��������̼����3��CO2+2NaOH=Na2CO3+H2O����4��80%����5���������ܻ�Ϊ���쵼�ܲ���ȼ��

��ϰ��ϵ�д�
 ������ÿ�ʱ�Ż���ҵϵ�д�
������ÿ�ʱ�Ż���ҵϵ�д�
�����Ŀ
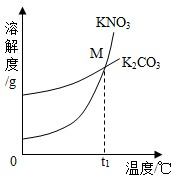 ��1��K2CO3��KNO3�ڲ�ͬ�¶�ʱ���ܽ�ȼ����ܽ��������ͼ��
��1��K2CO3��KNO3�ڲ�ͬ�¶�ʱ���ܽ�ȼ����ܽ��������ͼ��
| �¶�/�� | 20 | 30 | 50 | 60 | 80 | |
| �ܽ��/g | K2CO3 | 110 | 114 | 121 | 126 | 139 |
| KNO3 | 31.6 | 45.8 | 85.5 | 110 | 169 | |
��������M������______��
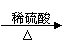
��2��ij����С�����÷�ͭ��ȡ����ͭ���������������������
����һ��Cu��CuO
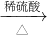 CuSO4
CuSO4����������Cu
 CuSO4[��֪Cu+2H2SO4��Ũ��
CuSO4[��֪Cu+2H2SO4��Ũ�� CuSO4+SO2��+2H2O]
CuSO4+SO2��+2H2O]����ɫ��ѧ�Ĺ۵����������Ϊ����______������������ԭ��______��
��1��K2CO3��KNO3�ڲ�ͬ�¶�ʱ���ܽ�ȼ����ܽ���������¡�
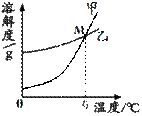 |
| �¶�/�� | 20 | 30 | 50 | 60 | 80 | |
| �ܽ��/g | K2CO3 | 110 | 114 | 121 | 126 | 139 |
| KNO3 | 31.6 | 45.8 | 85.5 | 110 | 169 |
��30��ʱ����124 g K2CO3�����м���100 gˮ������ܽ��������40�棬����Һ�����ʵ��������� ����������С�����䡱����
��������M������ ��
��2��ij����С�����÷�ͭ��ȡ����ͭ���������������������
| |||
 | |||
|
| |||
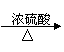 | |||
����������Cu CuSO4 [��֪Cu + 2H2SO4��Ũ���� CuSO4 + SO2��+2H2O]
����ɫ��ѧ�Ĺ۵����������Ϊ����_________������������ԭ��
��
��1��K2CO3��KNO3�ڲ�ͬ�¶�ʱ���ܽ�ȼ����ܽ��������ͼ��
��30��ʱ����124g K2CO3�����м���100gˮ������ܽ��������40�棬����Һ�����ʵ��������� ����������С�����䡱����
��������M������ ��
��2��ij����С�����÷�ͭ��ȡ����ͭ���������������������
����һ��Cu��CuO CuSO4
CuSO4
����������Cu CuSO4[��֪Cu+2H2SO4��Ũ��
CuSO4[��֪Cu+2H2SO4��Ũ�� CuSO4+SO2��+2H2O]
CuSO4+SO2��+2H2O]
����ɫ��ѧ�Ĺ۵����������Ϊ���� ������������ԭ�� ��
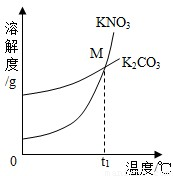
| �¶�/�� | 20 | 30 | 50 | 60 | 80 | |
| �ܽ��/g | K2CO3 | 110 | 114 | 121 | 126 | 139 |
| KNO3 | 31.6 | 45.8 | 85.5 | 110 | 169 | |
��������M������ ��
��2��ij����С�����÷�ͭ��ȡ����ͭ���������������������
����һ��Cu��CuO
 CuSO4
CuSO4����������Cu
 CuSO4[��֪Cu+2H2SO4��Ũ��
CuSO4[��֪Cu+2H2SO4��Ũ�� CuSO4+SO2��+2H2O]
CuSO4+SO2��+2H2O]����ɫ��ѧ�Ĺ۵����������Ϊ���� ������������ԭ�� ��

 ��2013?�ֿ���һģ��Cu��Zn�ĺϽ��Ϊ��ͭ���������ĵ����Ժ���ʴ�ԣ��������������������
��2013?�ֿ���һģ��Cu��Zn�ĺϽ��Ϊ��ͭ���������ĵ����Ժ���ʴ�ԣ��������������������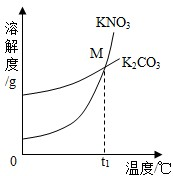 ��2013?��������ģ����1��K2CO3��KNO3�ڲ�ͬ�¶�ʱ���ܽ�ȼ����ܽ��������ͼ��
��2013?��������ģ����1��K2CO3��KNO3�ڲ�ͬ�¶�ʱ���ܽ�ȼ����ܽ��������ͼ��