��Ŀ����
 ��2013?�ֿ���һģ��Cu��Zn�ĺϽ��Ϊ��ͭ���������ĵ����Ժ���ʴ�ԣ��������������������
��2013?�ֿ���һģ��Cu��Zn�ĺϽ��Ϊ��ͭ���������ĵ����Ժ���ʴ�ԣ����������������������1���������ӳ��û�ͭ�Ƴɽ�������ġ���Ԫ������ƭ���ǣ����м����д�����ǣ�
A
A
������A������ɫ B�����ܶ�
C������������Һ D��������
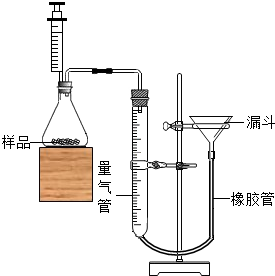
��2��Ϊ�˲ⶨijͭп�Ͻ����ɣ�У��ѧ����С�����øúϽ��ĩ��ϡ���ᷴӦ�����������ͼ��ʾ��ʵ��װ�ý���ʵ�飮
�ټ��װ�������Եķ����ǣ����Ӻ�װ�ã���©��עˮ�������������γɸ߶Ȳ���ñ�ǣ�һ��ʱ��߶Ȳ�
����
����
��˵�����������ã�����ȷ��ȡ��������Һ��ij�ʼ�����ն���ǰ������еIJ�����
��������Һ����ƽ
��������Һ����ƽ
���۸�С�����������ʵ�飬������ص�ʵ�����ݴ������£���ʵ���¶��¦�H2=0.09g/L��
| �Ͻ������/g | ϡ��������/mL | �������������/mL | |
| ��һ�� | 2 | 15 | 44.4 |
| �ڶ��� | 2 | 20 | 44.4 |
| ������ | 4 | 15 | 44.4 |
b�����ϱ����ݷ��������úϽ���ϡ�����������Ϊ
1��8
1��8
ʱ���Ͻ��е�п��ϡ����ǡ����ȫ��Ӧ��ϡ�����������������Ϊ1.21%
1.21%
����ʵ���¶��£���H2SO4=1.08g/cm3����������1������Cu��Zn��Au�Ļ�ѧ���ʺ��������ʷ�����ͭп�Ͻ��Ƴɵļٽ�Ԫ������Ȼ�������ƽ����죬���Ͻ��еĽ���ͭ��п��Զ��Ȼƽ�ǿ�����������Ƶ�����ѧ�������ϴ�ͭп�Ͻ������ᷢ���û���Ӧ���ų��������ڿ��������Ȼ��ɺ�ɫ�ȣ�������������Һ�����û���Ӧ��������Щ�����ϵIJ�����Ժܷ�����������ǣ�
��2���ٸ��ݳ���Һ������װ�����ܣ�װ���ڵ���ѹ���������ش�
�ڸ�����ͨ����ԭ����֪Ӧ�õ�������Һ����ƽ�ٽ��в������н��
�۷������������ݿ���֪����ͭ��п�Ͻ���ͭ��ϡ�����Ӧֻ��п��ϡ���ᷴӦ���ڶ��κ͵�һ����ȡ�ĺϽ�������ͬ���ڶ������õ�ϡ����ȵ�һ�ζ࣬���������ɵ�����������ͬ��˵��������ȡ�ĺϽ��е�п����ȫ��Ӧ�ˣ��ɴ˿���֪��2g�Ͻ�ֻ������44.4mL��������������ȡ�ĺϽ���ǰ���ε�2��������Ͻ��е�п��ȫ��Ӧ��Ӧ�ò���88.8mL���壬��ʵ���ϲ����������ǰ������ͬ��˵�������η�Ӧ�кϽ��������ϡ���ᷴӦ��ȫ���ɴ˿���֪��15mlϡ����ֻ�ܲ���44.4mL�������Ӷ������жϳ�ϡ����ͺϽ�ǡ����ȫ��Ӧ�������ȣ������������������Լ����п�������������������������Լ����15mlϡ���������ʵ��������Ӷ������ϡ���������������
��2���ٸ��ݳ���Һ������װ�����ܣ�װ���ڵ���ѹ���������ش�
�ڸ�����ͨ����ԭ����֪Ӧ�õ�������Һ����ƽ�ٽ��в������н��
�۷������������ݿ���֪����ͭ��п�Ͻ���ͭ��ϡ�����Ӧֻ��п��ϡ���ᷴӦ���ڶ��κ͵�һ����ȡ�ĺϽ�������ͬ���ڶ������õ�ϡ����ȵ�һ�ζ࣬���������ɵ�����������ͬ��˵��������ȡ�ĺϽ��е�п����ȫ��Ӧ�ˣ��ɴ˿���֪��2g�Ͻ�ֻ������44.4mL��������������ȡ�ĺϽ���ǰ���ε�2��������Ͻ��е�п��ȫ��Ӧ��Ӧ�ò���88.8mL���壬��ʵ���ϲ����������ǰ������ͬ��˵�������η�Ӧ�кϽ��������ϡ���ᷴӦ��ȫ���ɴ˿���֪��15mlϡ����ֻ�ܲ���44.4mL�������Ӷ������жϳ�ϡ����ͺϽ�ǡ����ȫ��Ӧ�������ȣ������������������Լ����п�������������������������Լ����15mlϡ���������ʵ��������Ӷ������ϡ���������������
����⣺��1���⣺A���۲���ɫ�����ڶ��ߵ���ɫ����һ�£��������ڱȽ�Ϊ��٣��ʴ���
B������ܶȱ�ͭ��п�Ķ�������ͭп�Ͻ���ܶ���ȻС�ڽ���ܶȣ���˿���ͨ�������������ܶȼ�����α��
C�������������У�ͭп��������ǰ�棬���û����������е�����������һ������ɫ������Ϊ�٣���˿������ý������û���Ӧ������α��
D�����������У�п�����ᷴӦ������������������Ӧ�����������ų���Ϊ�٣����������������α��
��ѡ��Ϊ��A��
��2���ټ��װ�������Եķ����ǣ����Ӻ�װ�ã���©��עˮ�������������γɸ߶Ȳ���ñ�ǣ�һ��ʱ��߶Ȳ�䣬˵�����������ã�
�ڶ�ȡ������������Һ��̶�ǰ����Ҫ���еIJ����ǵ��������ܸ߶ȣ�ʹ����Һ����ƽ��������е�Һ���ܲ���ѹǿ���Թ��е��������ѹ�������죬Ӱ�����������ⶨ�������ȷ��ȡ��������Һ��ij�ʼ�����ն���ǰ������еIJ����ǵ�������Һ����ƽ��
�۱Ƚ��������ݿ���֪����2g�Ͻ�ֻ������44.4mL���������ɵ����ο�֪����44.4mL����ֻ����15mlϡ���ᣬ����2g�Ͻ��е�п��15mlϡ�����ǡ����ȫ��Ӧ����44.4mL��44.4mL��1000mL/L��0.09g/L=0.004g��������һ�κϽ��ϡ����ǡ����ȫ��Ӧ��������úϽ���ϡ�����������Ϊ��2g����15mL��1.08g/cm3��=1��8��
��2gͭп�Ͻ��к���п������Ϊx����Ӧ���������Ϊy
Zn+H2SO4�TZnSO4+H2��
65 98 2
x y 0.004g
=
x=0.13g
=
y=0.196g
a��ͭп�Ͻ���ͭ����������=
��100%=93.5%
b�����������������
��100%=1.21%��
��ͭп�Ͻ���ͭ����������Ϊ 93.5%��ϡ�����������������Ϊ1.21%��
�ʴ�Ϊ����1��A ��2���ٲ��� �ڵ�������Һ����ƽ
����2gͭп�Ͻ��к���п������Ϊx����Ӧ���������Ϊy
Zn+H2SO4�TZnSO4+H2��
65 98 2
x y 0.004g
=
x=0.13g
=
y=0.196g
a��ͭп�Ͻ���ͭ����������=
��100%=93.5%
b�����������������
��100%=1.21%��
��ͭп�Ͻ���ͭ����������Ϊ 93.5%��ϡ�����������������Ϊ1.21%��
��ͭп�Ͻ���ͭ����������Ϊ 35%����һ��ʵ�飬�Ͻ��е�п��ϡ����ǡ����ȫ��Ӧ��������Ϊ1��8��ϡ�����������������Ϊ1.21%��
B������ܶȱ�ͭ��п�Ķ�������ͭп�Ͻ���ܶ���ȻС�ڽ���ܶȣ���˿���ͨ�������������ܶȼ�����α��
C�������������У�ͭп��������ǰ�棬���û����������е�����������һ������ɫ������Ϊ�٣���˿������ý������û���Ӧ������α��
D�����������У�п�����ᷴӦ������������������Ӧ�����������ų���Ϊ�٣����������������α��
��ѡ��Ϊ��A��
��2���ټ��װ�������Եķ����ǣ����Ӻ�װ�ã���©��עˮ�������������γɸ߶Ȳ���ñ�ǣ�һ��ʱ��߶Ȳ�䣬˵�����������ã�
�ڶ�ȡ������������Һ��̶�ǰ����Ҫ���еIJ����ǵ��������ܸ߶ȣ�ʹ����Һ����ƽ��������е�Һ���ܲ���ѹǿ���Թ��е��������ѹ�������죬Ӱ�����������ⶨ�������ȷ��ȡ��������Һ��ij�ʼ�����ն���ǰ������еIJ����ǵ�������Һ����ƽ��
�۱Ƚ��������ݿ���֪����2g�Ͻ�ֻ������44.4mL���������ɵ����ο�֪����44.4mL����ֻ����15mlϡ���ᣬ����2g�Ͻ��е�п��15mlϡ�����ǡ����ȫ��Ӧ����44.4mL��44.4mL��1000mL/L��0.09g/L=0.004g��������һ�κϽ��ϡ����ǡ����ȫ��Ӧ��������úϽ���ϡ�����������Ϊ��2g����15mL��1.08g/cm3��=1��8��
��2gͭп�Ͻ��к���п������Ϊx����Ӧ���������Ϊy
Zn+H2SO4�TZnSO4+H2��
65 98 2
x y 0.004g
| 65 |
| x |
| 2 |
| 0.004g |
| 98 |
| y |
| 2 |
| 0.004g |
a��ͭп�Ͻ���ͭ����������=
| 2g-0.13g |
| 2g |
b�����������������
| 0.196g |
| 15ml��1.08g/ml |
��ͭп�Ͻ���ͭ����������Ϊ 93.5%��ϡ�����������������Ϊ1.21%��
�ʴ�Ϊ����1��A ��2���ٲ��� �ڵ�������Һ����ƽ
����2gͭп�Ͻ��к���п������Ϊx����Ӧ���������Ϊy
Zn+H2SO4�TZnSO4+H2��
65 98 2
x y 0.004g
| 65 |
| x |
| 2 |
| 0.004g |
| 98 |
| y |
| 2 |
| 0.004g |
a��ͭп�Ͻ���ͭ����������=
| 2g-0.13g |
| 2g |
b�����������������
| 0.196g |
| 15ml��1.08g/ml |
��ͭп�Ͻ���ͭ����������Ϊ 93.5%��ϡ�����������������Ϊ1.21%��
��ͭп�Ͻ���ͭ����������Ϊ 35%����һ��ʵ�飬�Ͻ��е�п��ϡ����ǡ����ȫ��Ӧ��������Ϊ1��8��ϡ�����������������Ϊ1.21%��
���������⿼����ǺϽ�����ʼ����ݻ�ѧ����ʽ���йؼ��㣬ѧ�����ÿ��Ʊ���������ȷ�ķ����������е������ǽ�����Ĺؼ���

��ϰ��ϵ�д�
�����Ŀ
