��Ŀ����
2��һ���Ȼ�þ����������ɵĻ�����У�Ϊ�ⶨ�����Ȼ�þ������������С��ͬѧ��ȡ�û������ Ʒ 20g����ȫ����ˮ�У�Ȼ��ȡ����һ��������������������������Һ 100g��ƽ�����Ĵμ������У������ ���ʵ���������±���ʾ���������������صļ��㣺| ���� | 1 | 2 | 3 | 4 |
| ��������������Һ��������g�� | 25 | 25 | 25 | 25 |
| ���ɳ�����������g�� | 2.9 | X | 8.7 | 8.7 |
��2������ԭ����������Ʒ���Ȼ�þ������������71.25%��
���� ��1��ÿ����25g����������Һ��ȫ��Ӧ�����ɳ���2.9g����ˣ���2�μ����25g����������Һ��ȫ��Ӧ�������ɳ���2.9g��
��2�����Ȼ�þ�����������������ȫ��Ӧ�����ɳ���������þ8.7g�����ݷ�Ӧ�Ļ�ѧ����ʽ���������Ʒ���Ȼ�þ��������Ȼ������Ʒ������ȿɵû������Ʒ���Ȼ�þ������������
��� �⣺��1��ÿ����25g����������Һ��ȫ��Ӧ�����ɳ���2.9g����ˣ���2�μ���25g����������Һ��ȫ��Ӧ�����ó�����������=2.9g��2=5.8g
�ʴ�Ϊ��5.8��
��2�����������Ʒ���Ȼ�þ������Ϊx
MgCl2+2NaOH�TMg��OH��2��+2NaCl
95 58
x 8.7g
$\frac{95}{x}=\frac{58}{8.7g}$
x=14.25g
ԭ����������Ʒ���Ȼ�þ����������=$\frac{14.25g}{20g}$��100%=71.25%
�𰸣�
��1��5.8g
��2��71.25%
���� ������Ҫ����ѧ�����û�ѧ����ʽ���м�����ƶϵ�����������ʱҪע��淶�Ժ�ȷ�ԣ�

��ϰ��ϵ�д�
�����Ŀ
13���ӽ������õ���ʷ������������ͭ��ʱ��������������ʱ�������������ǽ�������£�����Ⱥ�˳����Ҫ�������йص��ǣ�������
�ٵؿ��еĽ���Ԫ�صĺ������ڽ������˳�۽����ĵ����ԣ��ܽ���ұ�������׳̶ȣ��ݽ�������չ�ԣ�
�ٵؿ��еĽ���Ԫ�صĺ������ڽ������˳�۽����ĵ����ԣ��ܽ���ұ�������׳̶ȣ��ݽ�������չ�ԣ�
| A�� | �٢� | B�� | �ڢ� | C�� | �ۢ� | D�� | �ڢ� |
17�����������ʵ���Һ����Ba��OH��2����Na2SO4����HNO3����CuCl2�����������Լ��Ϳɽ�������һ��������������˳���ǣ�������
| A�� | �ܢ٢ڢ� | B�� | �٢ۢܢ� | C�� | �ܢۢڢ� | D�� | �٢ܢڢ� |
6��2013��4��25����8�㣬̨����·�����彭�ֵ�һ�����ѳ���ʮ��ֽ���þ������ȼ������þ��һ������ɫ�Ľ�����ȼ��ʱ�ܲ���ѣĿ�İ⣬��������ը�����⣬����þ�ڸ�������������ˮ���ͷų������������ڶ�����̼�о���ȼ�ղ���һ�ְ�ɫ��ĩ��һ�ֺ�ɫ��ĩ������Ϊ���Ľ��۲���ȷ���ǣ�������
| A�� | ijЩ������ȼ�ղ�����CO2��� | |
| B�� | �÷�Ӧ�Ƿų����� | |
| C�� | �÷�Ӧ��ʵ����Mg��CO2�л��е�O2��Ӧ | |
| D�� | ijЩ������ȼ�ղ�����ˮ�˾� |
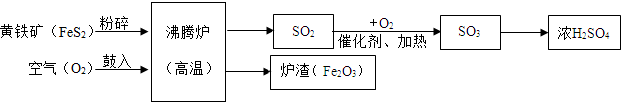
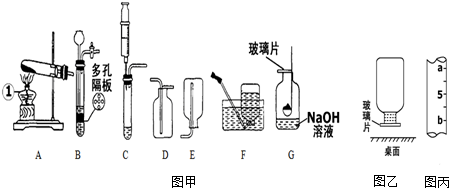
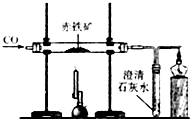 ����������⡢̽������ͽ��������ѧ�û�ѧ����Ҫ������ij�о���ѧϰС���ͬѧ��ѧϰ�����仯����֪ʶʱ������ϵ���о���
����������⡢̽������ͽ��������ѧ�û�ѧ����Ҫ������ij�о���ѧϰС���ͬѧ��ѧϰ�����仯����֪ʶʱ������ϵ���о���