��Ŀ����
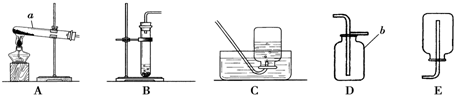
�������ͼʾװ�ã��ش��й����⣺
��1������a�������� ��
��2��ʵ�����ø��������ȡ��������ѡ��ķ���װ���� �����ţ����Թܿڷ�һ������������ ����װ��C�ռ����������ܿڿ�ʼ�����ݷų�ʱ�����������ռ��������� �����������ļ���ƿӦ ��������������������ϣ�
��3��ʵ���ҳ���ʯ��ʯ��ϡ���ᷴӦ��ȡ������̼���䷴Ӧ�Ļ�ѧ����ʽΪ �������ſ������ռ�������̼���ռ�װ��Ӧѡ�� �����ţ���
���𰸡���������1��ֱ��д�����������ƣ�
��2������ʵ�������ø��������ȡ����ķ�Ӧ��״̬�ͷ�Ӧ����ѡ����װ�ã�����ע����������𣬲����������ܶ�ȷ������ƿ�ķ��÷�����
��3������ʵ��������ȡ������̼�ķ�Ӧԭ����д����ʽ�����ݶ�����̼���ܶ�ѡ���ռ�װ�ã�
����⣺��1��ֱ��д������������Ϊ���ƾ��ƣ�
��2����������ڼ��ȵ������¿��Էֽ�����������̡�����غ����������ڹ�������ͣ���ѡ����װ��A��������سʷ�ĩ״���ڼ���ʱ�����뵼�ܶ��������ܣ�����Ҫ���Թܿڷ�һ�����������Թ����п��������Ըտ�ʼ����ʱ�����������ʹӶ��ݳ��Թܣ���ʱ���ռ����ʹ�ռ��������岻�����������ܶȱȿ������ռ��������ļ���ƿӦ���������ϣ�
��3��ʯ��ʯ��ϡ���ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼������ʽ�ǣ�CaCO3+2HCl�TCaCl2+H2O+CO2����������̼�ܶȱȿ���������Ӧ��ѡ��Bװ�����ռ�������̼��
�ʴ�Ϊ����1���ƾ���
��2��A�� ��ֹ����ʱ������ط�ĩ���뵼�ܣ� �ռ��������л��п����� ����
��3��CaCO3+2HCl�TCaCl2+H2O+CO2���� B��
��������������ʵ��������ȡ����ķ���װ�ú��ռ�װ�õ�ѡ�����ݣ�����������ȷ��ѡ��ס����������ȡ�ķ�Ӧԭ�������ܹ���д��ѧ����ʽ����ȷʵ������ȡ����ʱ��ע�����
��2������ʵ�������ø��������ȡ����ķ�Ӧ��״̬�ͷ�Ӧ����ѡ����װ�ã�����ע����������𣬲����������ܶ�ȷ������ƿ�ķ��÷�����
��3������ʵ��������ȡ������̼�ķ�Ӧԭ����д����ʽ�����ݶ�����̼���ܶ�ѡ���ռ�װ�ã�
����⣺��1��ֱ��д������������Ϊ���ƾ��ƣ�
��2����������ڼ��ȵ������¿��Էֽ�����������̡�����غ����������ڹ�������ͣ���ѡ����װ��A��������سʷ�ĩ״���ڼ���ʱ�����뵼�ܶ��������ܣ�����Ҫ���Թܿڷ�һ�����������Թ����п��������Ըտ�ʼ����ʱ�����������ʹӶ��ݳ��Թܣ���ʱ���ռ����ʹ�ռ��������岻�����������ܶȱȿ������ռ��������ļ���ƿӦ���������ϣ�
��3��ʯ��ʯ��ϡ���ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼������ʽ�ǣ�CaCO3+2HCl�TCaCl2+H2O+CO2����������̼�ܶȱȿ���������Ӧ��ѡ��Bװ�����ռ�������̼��
�ʴ�Ϊ����1���ƾ���
��2��A�� ��ֹ����ʱ������ط�ĩ���뵼�ܣ� �ռ��������л��п����� ����
��3��CaCO3+2HCl�TCaCl2+H2O+CO2���� B��
��������������ʵ��������ȡ����ķ���װ�ú��ռ�װ�õ�ѡ�����ݣ�����������ȷ��ѡ��ס����������ȡ�ķ�Ӧԭ�������ܹ���д��ѧ����ʽ����ȷʵ������ȡ����ʱ��ע�����

��ϰ��ϵ�д�
 ��������ϵ�д�
��������ϵ�д�
�����Ŀ