��Ŀ����
Ϊ�˲ⶨ��ͭм����п��ͭ�ֳɵĺϽ���Ʒ����ɣ��ס��ҡ���������λͬѧ�ֱ�ȡ����ϡ���ᷴӦ��ϡ������ܶ�Ϊ1.17g/mL����ʵ�����ݼ�¼���£�
| �� | �� | �� | �� | |
| ȡ�û�ͭм������/g | 8.0 | 8.0 | 8.0 | 12.0 |
| ȡ��ϡ��������/mL | 8.0 | 16.0 | 24.0 | һ����� |
| ��������������/g | 6.7 | 5.4 | 4.8 | 8.1 |
��1��8.0g��ͭм��п��������______
��2����ͬѧ����ϡ����������______
��3����ͬѧ���������Һ�����ʵ�����������______��
�⣺��1����ʵ���֪������8.0mLϡ����ʱ�����Ľ�������=8.0g-6.7g=1.3g����˵�����24.0mLϡ����ʱ�������Ľ���������=1.3g�� =3.9g������ʵ������Ʒ��������=8.0g-4.8g=3.2g��3.9g����ˣ���ʵ����Ʒ��п����ȫ��Ӧ����������Ϊͭ����8.0g��ͭм��п������Ϊ3.2g��
=3.9g������ʵ������Ʒ��������=8.0g-4.8g=3.2g��3.9g����ˣ���ʵ����Ʒ��п����ȫ��Ӧ����������Ϊͭ����8.0g��ͭм��п������Ϊ3.2g��
��2����ʵ���֪������8.0mLϡ����ʱ�����Ľ�������=8.0g-6.7g=1.3g��ʵ�鶡�����Ľ�������=12.0g-8.1g=3.9g����������ϡ��������= ��8.0mL=24mL��
��8.0mL=24mL��
��3�����ͬѧ��ʵ������������п������Ϊx���ų�����������Ϊy
Zn+H2SO4�TZnSO4+H2��
65 161 2
1.3g x y
 =
= =
=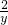
x=3.22g��y=0.04g
��ͬѧ���������Һ�����ʵ���������= ��100%=30.3%
��100%=30.3%
�ʴ�Ϊ����1��3.2g����2��24ml����3��30.3%��
��������1���Աȼס��ҡ�������ʵ������ϡ����ijɱ����ӣ��������������ı仯���ɵó�ʵ�����Ʒ�н���п����ȫ��Ӧ����������Ϊͭ���������Ʒ��п��������
��2����ʵ��ɵ���Ʒÿ����1.3g����ϡ����8.0mL����˿���ʵ�鶡��Ʒ�����ļ���������������ϡ����������
��3��ʵ�����ϡ������ȫ��Ӧ�γ�����п��Һ�������Ľ���п�����������ݷ�Ӧ�Ļ�ѧ����ʽ�������������п�������������������غ㶨����÷�Ӧ����Һ������������÷�Ӧ����Һ����������������
��������ʵ��õ���Ӧ��ϡ��������������Ʒ��п��������ϵ����Ϊ�������Ļ��������ַ��������������
 =3.9g������ʵ������Ʒ��������=8.0g-4.8g=3.2g��3.9g����ˣ���ʵ����Ʒ��п����ȫ��Ӧ����������Ϊͭ����8.0g��ͭм��п������Ϊ3.2g��
=3.9g������ʵ������Ʒ��������=8.0g-4.8g=3.2g��3.9g����ˣ���ʵ����Ʒ��п����ȫ��Ӧ����������Ϊͭ����8.0g��ͭм��п������Ϊ3.2g����2����ʵ���֪������8.0mLϡ����ʱ�����Ľ�������=8.0g-6.7g=1.3g��ʵ�鶡�����Ľ�������=12.0g-8.1g=3.9g����������ϡ��������=
 ��8.0mL=24mL��
��8.0mL=24mL����3�����ͬѧ��ʵ������������п������Ϊx���ų�����������Ϊy
Zn+H2SO4�TZnSO4+H2��
65 161 2
1.3g x y
 =
= =
=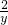
x=3.22g��y=0.04g
��ͬѧ���������Һ�����ʵ���������=
 ��100%=30.3%
��100%=30.3%�ʴ�Ϊ����1��3.2g����2��24ml����3��30.3%��
��������1���Աȼס��ҡ�������ʵ������ϡ����ijɱ����ӣ��������������ı仯���ɵó�ʵ�����Ʒ�н���п����ȫ��Ӧ����������Ϊͭ���������Ʒ��п��������
��2����ʵ��ɵ���Ʒÿ����1.3g����ϡ����8.0mL����˿���ʵ�鶡��Ʒ�����ļ���������������ϡ����������
��3��ʵ�����ϡ������ȫ��Ӧ�γ�����п��Һ�������Ľ���п�����������ݷ�Ӧ�Ļ�ѧ����ʽ�������������п�������������������غ㶨����÷�Ӧ����Һ������������÷�Ӧ����Һ����������������
��������ʵ��õ���Ӧ��ϡ��������������Ʒ��п��������ϵ����Ϊ�������Ļ��������ַ��������������

��ϰ��ϵ�д�
�����Ŀ
ijͬѧΪ�˲ⶨ��ͭм����п��ͭ�γɵĺϽ���Ʒ��ɣ����Ĵ�ȡ��Ʒ��ϡ���ᷴӦ����ʵ�����ݼ�¼���±���
�Լ��㣺��1����ͭ��п������������
��2������ϡ���������ʵ�����������
| 1 | 2 | 3 | 4 | |
| ȡ��Ʒ������g�� | 50.0 | 50.0 | 50.0 | 50.0 |
| ȡϡ����������g�� | 40.0 | 80.0 | 120.0 | 160.0 |
| ��������������g�� | 0.4 | 0.8 | 1.0 | 1.0 |
��2������ϡ���������ʵ�����������
ijͬѧΪ�˲ⶨ��ͭм����п��ͭ�γɵĺϽ���Ʒ����ɣ�ȡ�ķ���Ʒ�ֱ��ϡ���ᷴӦ����ʵ�����ݼ�¼���±���
�Լ��㣺
��1�����������ڵ�2����Ʒ��õ������У� �������ʣ���ȫ��Ӧ�ˣ�
��2����ʽ��������ϡ���������ʵ�����������
��3��������ͼ�л�����50.0g��Ʒ�м�ϡ���������������������仯��ϵ��ʾ��ͼ��

| �� Ʒ | ��1�� | ��2�� | ��3�� | ��4�� |
| ȡ��Ʒ������g�� | 50.0 | 50.0 | 50.0 | 50.0 |
| ȡϡ����������g�� | 40.0 | 80.0 | 120.0 | 160.0 |
| ��������������g�� | 0.4 | 0.8 | 1.0 | 1.0 |
��1�����������ڵ�2����Ʒ��õ������У�
��2����ʽ��������ϡ���������ʵ�����������
��3��������ͼ�л�����50.0g��Ʒ�м�ϡ���������������������仯��ϵ��ʾ��ͼ��

ijͬѧΪ�˲ⶨ��ͭм����п��ͭ�γɵĺϽ���Ʒ��ɣ�ȡ�ķ���Ʒ�ֱ��ϡ���ᷴӦ����ʵ�����ݼ�¼���±���
�Լ��㣺
��1�����ķ���Ʒ����ʵ���У�ϡ������ʣ����� ��
��2����ͭм��ϡ����ǡ����ȫ��Ӧʱ��������Ϊ ��
��3����ʽ�����ͭ��Ʒ��ͭ������������
| ��Ʒ | ��1�� | ��2�� | ��3�� | ��4�� |
| ȡ��Ʒ������g�� | 50.0 | 50.0 | 50.0 | 50.0 |
| ȡϡ����������g�� | 40.0 | 80.0 | 120.0 | 160.0 |
| ��������������g�� | 0.4 | 0.8 | 1.0 | 1.0 |
��1�����ķ���Ʒ����ʵ���У�ϡ������ʣ�����
��2����ͭм��ϡ����ǡ����ȫ��Ӧʱ��������Ϊ
��3����ʽ�����ͭ��Ʒ��ͭ������������