��Ŀ����
ʵ�����е��Լ�һ��Ҫ�ܷⱣ�棬������ܻ�������Ӵ������ʡ�ij�о���ѧϰС�鷢��һƿδ�ܱյ�KOH���壬����ɷ�������¼��裬�������ʵ��̽����
����1��ֻ��KOH��
����2����KOH��K2CO3�� ����3��ֻ��K2CO3
��1���ɷ��п��ܺ���K2CO3��ԭ���ǣ��û�ѧ����ʽ�ش�_______________________��
��2��ȡ������Ʒ���Թ��У���������ϡ���ᣬ�۲쵽_____________________��˵������2�����3������
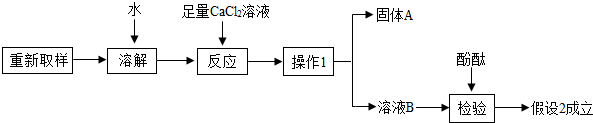
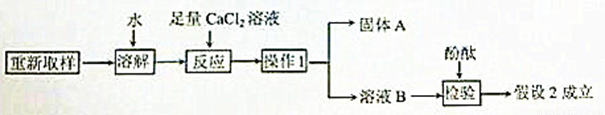
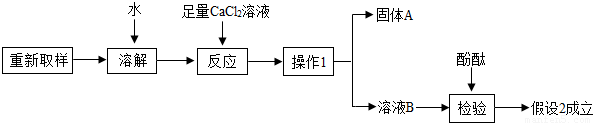
��3����һ��̽���Ĺ������£�
����1��ֻ��KOH��
����2����KOH��K2CO3�� ����3��ֻ��K2CO3
��1���ɷ��п��ܺ���K2CO3��ԭ���ǣ��û�ѧ����ʽ�ش�_______________________��
��2��ȡ������Ʒ���Թ��У���������ϡ���ᣬ�۲쵽_____________________��˵������2�����3������
��3����һ��̽���Ĺ������£�

�١�����1����������____________����
�ڡ�����A���Ļ�ѧʽ��___________��
�ۼ�������CaCl2��Һ�������� _______________________________��
��4�������Լ�Ҳ�����ü�ֵ����KOH��K2CO3����ɺ����ʿ�����ƿ�����Լ�����;��______________��д��һ������
�ڡ�����A���Ļ�ѧʽ��___________��
�ۼ�������CaCl2��Һ�������� _______________________________��
��4�������Լ�Ҳ�����ü�ֵ����KOH��K2CO3����ɺ����ʿ�����ƿ�����Լ�����;��______________��д��һ������
(1)2KOH + CO2 === K2CO3 + H2O
(2) �����ݲ���
(3) �ٹ���
��CaCO3
�� ��̼�����ȫ��Ӧ����ֹ�Խ�������ʵ�����
(4) ���طʣ������𰸺�����ɣ�
(2) �����ݲ���
(3) �ٹ���
��CaCO3
�� ��̼�����ȫ��Ӧ����ֹ�Խ�������ʵ�����
(4) ���طʣ������𰸺�����ɣ�

��ϰ��ϵ�д�
 ֥�鿪���γ�������ϵ�д�
֥�鿪���γ�������ϵ�д�
�����Ŀ