��Ŀ����
��������ɽˮ�೧����ʡ��ģ�ϴ��ˮ�೧���ó��Ļ�����Ϊ�˲ⶨij��ɽʯ��ʯ��̼��Ƶ�����������ȡʯ��ʯ��Ʒ��������10%��ϡ�������ձ��з�Ӧ������ʯ��ʯ��Ʒ�����ʲ���ϡ���ᷴӦҲ������ˮ�����й�ʵ���������±���
| ��Ӧǰ | ��Ӧ�� | ||
| ʵ������ | �ձ���ϡ��������� | ʯ��ʯ��Ʒ������ | �ձ������л��������� |
| 134.4g | 10g | 141.1g | |
��1����ʯ��ʯ��̼��Ƶ�����������
��2��������10%�������������
���ʯ��ʯ��Ʒ��̼��Ƶ�����Ϊx�����ĵ��������ʵ�����Ϊy
CaCO3+2HCl=CaCl2+H2O+CO2��
100 73 44
x y 3.3g
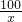 =
= x=7.5g
x=7.5g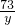 =
= y=5.475g
y=5.475g��ʯ��ʯ��̼��Ƶ���������Ϊ��
 ��100%=75%
��100%=75%��2�������ĵ�10%�������������5.475g��10%=54.75g
�𣺸�ʯ��ʯ��̼��Ƶ���������Ϊ75%��������10%�������������54.75g��
���������������غ㶨�ɿ�֪����Ӧǰ���������Ϊ���ɶ�����̼����������֪������̼�����������ݻ�ѧ����ʽ������μӷ�Ӧ��̼��Ƶ��������������ʵ��������Ӷ�����ʯ��ʯ��̼��Ƶ����������������ʵ����������ɼ���������ĵ������������
���������������û�ѧ�еĻ���˼�������غ�˼�룬ͨ�����������Ļ�ѧ��Ӧԭ����֪���ɵĶ�����̼�������������з���ʽ���㣬�Ӷ�������ѧ���ķ�������������������

��������ɽˮ�೧����ʡ��ģ�ϴ��ˮ�೧���ó��Ļ�����Ϊ�˲ⶨij��ɽʯ��ʯ��̼��Ƶ�����������ȡʯ��ʯ��Ʒ��������10%��ϡ�������ձ��з�Ӧ������ʯ��ʯ��Ʒ�����ʲ���ϡ���ᷴӦҲ������ˮ�����й�ʵ���������±���
|
|
��Ӧǰ |
��Ӧ�� |
|
|
ʵ������ |
�ձ���ϡ��������� |
ʯ��ʯ��Ʒ������ |
�ձ������л��������� |
|
134.4g |
10g |
141.1g |
����㣨д��������̣���
��1����ʯ��ʯ��̼��Ƶ�����������
��2��������10%�������������
��������ɽˮ�೧����ʡ��ģ�ϴ��ˮ�೧���ó��Ļ�����Ϊ�˲ⶨij��ɽʯ��ʯ��̼��Ƶ�����������ȡʯ��ʯ��Ʒ��������10%��ϡ�������ձ��з�Ӧ������ʯ��ʯ��Ʒ�����ʲ���ϡ���ᷴӦҲ������ˮ�����й�ʵ���������±���
|
|
��Ӧǰ |
��Ӧ�� |
|
|
ʵ������ |
�ձ���ϡ��������� |
ʯ��ʯ��Ʒ������ |
�ձ������л��������� |
|
134.4g |
10g |
141.1g |
����㣨д��������̣���
��1����ʯ��ʯ��̼��Ƶ�����������
��2��������10%�������������
��������ɽˮ�೧����ʡ��ģ�ϴ��ˮ�೧���ó��Ļ�����Ϊ�˲ⶨij��ɽʯ��ʯ��̼��Ƶ�����������ȡʯ��ʯ��Ʒ��������10%��ϡ�������ձ��з�Ӧ������ʯ��ʯ��Ʒ�����ʲ���ϡ���ᷴӦҲ������ˮ�����й�ʵ���������±���
| ��Ӧǰ | ��Ӧ�� |
| |
| ʵ������ | �ձ���ϡ��������� | ʯ��ʯ��Ʒ������ | �ձ������л��������� |
| 134.4g | 10g | 141.1g |
����㣨д��������̣���
��1����ʯ��ʯ��̼��Ƶ�����������
��2��������10%�������������