��Ŀ����
ij��ȤС���ͬѧ��ʵ�����ú�������CaCl2��NaCl��������һ������������NaCl��Һ��ʵ�鲽����ͼ��ʾ��
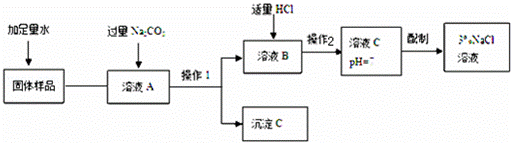
��ش�
��1������C�Ļ�ѧʽΪ������
��2�����롰����Na2CO3����������Ӧ�Ļ�ѧ����ʽΪ������
��3���ڡ�����1���õ�ʵ�鷽����������
��4�����롰����HCl����Ŀ����������
��5��������2����Ŀ���Dzⶨ��ҺΪ���ԣ��ⶨ��ҺpH�ķ��������ڡ���
��6�����ⶨ����ҺC��������Ϊ50g����������Ϊ6%���Ȼ�����Һ���ܶ�Ϊ1.04g/cm3������Ҫ�ø���Һ����50g��������Ϊ3%���Ȼ�����Һ��������Ȼ�����Һ��ˮ������ֱ�Ϊ����ml������ml����ˮ�ܶ�Ϊ1.0g/cm3����
���ƹ����г�Ҫ�õ����������⣬����Ҫ�IJ���������������
| ���ȷ�������ͼ�����ʵ��������������ʵĻ�ѧ���ʽ��з������ ��1����̼���ƺ��Ȼ��Ʒ�Ӧ����̼��Ƴ����ش� ��2����̼���ƺ��Ȼ��Ʒ�Ӧԭ����д����ʽ�� ��3������1�ǹ��˲����������Ƿ�����ܡ����������ʵķ����� ��4����Ϊ��ҺA�����ӵ�Na2CO3��Һ�ǹ����ģ�������ҺB����ʣ���Na2CO3�����ɵ�NaCl������������������Ϊ�˳�ȥNa2CO3��ͬʱ�������µ����ʣ�ʹ��Һ�����ԣ���5����pH��ֽ�ⶨ��ҺpHֵ�ķ����ش� ��6�����ʵ�����=��Һ�����������ʵ�������������Һϡ��ǰ�����ʵ��������䣬������Һ��Ҫ�IJ�������������Ͳ����ͷ�ιܣ� | |
| ��� | �⣺��1��̼���ƺ��Ȼ��Ʒ�Ӧ����̼��Ƴ����� ��2��̼���ƺ��Ȼ��Ʒ�Ӧ����̼��Ƴ������Ȼ��ƣ�����ʽ��CaCl2+Na2CO3═CaCO3��+2NaCl�� ��3��ͨ������1�������Һ����룬�����˹��˵ķ����� ��4����������ϡ�����Ŀ����Ϊ�˳�ȥ����1�й�����̼���ƣ�ͬʱʹ��Һ�����ԣ� ��5���ⶨ��ҺpH�ķ������ڸ���IJ���Ƭ�Ϸ�һƬpH��ֽ���ø���IJ�����պȡ����Һ����pH��ֽ�ϣ�Ȼ����ʾ����ɫ�����ɫ�����գ�������Һ��pHֵ�� ��6������Ҫ6%�Ȼ�����Һ��������x����x��6%=50g��3%�����x=25g������Ҫ�Ȼ�����Һ�����Ϊ�� �ʴ�Ϊ����1��CaCO3�� ��2��CaCl2+Na2CO3=CaCO3��+2NaCl�� ��3�����ˣ� ��4������ҺB������ Na2CO3 ȫ��ת��ΪNaCl������ҺΪ���ԣ� ��5���ڸ��ﲣ��Ƭ�Ϸ�һƬpH��ֽ���ø��ﲣ����պȡ��Һ������pH��ֽ�ϣ�����ֽ��ʾ����ɫ���ɫ�����գ�ȷ����Һ��pHֵ�� ��6��24�� 25�� ��Ͳ�� ��ͷ�ιܣ� |
��

| ʵ �� �� �� | 1 | 2 | 3 | 4 | 5 |
| ����ϡ���������/g | 20.0 | 20.0 | 20.0 | 20.0 | 20.0 |
| ʣ����������/g | 6.00 | m | 2.00 | 1.20 | 1.20 |
��l������m��ֵΪ
��2����ʯ��ʯ��Ʒ�Ĵ��ȣ�
��3�����õ�ϡ���������ʵ�����������
��4����2��ʵ���������Һ�����ʵ�����������
ijУ��ȤС���ͬѧ���ۺ�ʵ����У���ˮ�೧����һ��ʯ��ʯ��Ʒ������Ա�������Ǵ���Ʒ�к��е������Ƕ������裬��������������ˮ�����������ᷴӦ��Ϊ�˲ⶨ��ʯ��ʯ�Ĵ��ȣ���ȤС���ͬѧȡ��8.00g����ʯ��ʯ��Ʒ����ʵ�������е�δ֪��������������ϡ����100�˷�5�μ��룬��ַ�Ӧ�����ˡ�����Ȳ�����������õ��������ݣ�
| ʵ �� �� �� | 1 | 2 | 3 | 4 | 5 |
| ����ϡ���������/g | 20.0 | 20.0 | 20.0 | 20.0 | 20.0 |
| ʣ����������/g | 6.00 | m | 2.00 | 1.20 | 1.20 |
�Իش��������⣺
��l������m��ֵΪ_________g��
(2����ʯ��ʯ��Ʒ�Ĵ��ȡ�
��3�����õ�ϡ���������ʵ�����������
��4����2��ʵ���������Һ�����ʵ�����������
| ʵ �� �� �� | 1 | 2 | 3 | 4 | 5 |
| ����ϡ���������/g | 20.0 | 20.0 | 20.0 | 20.0 | 20.0 |
| ʣ����������/g | 6.00 | m | 2.00 | 1.20 | 1.20 |
��l������m��ֵΪ______g��
��2����ʯ��ʯ��Ʒ�Ĵ��ȣ�
��3�����õ�ϡ���������ʵ�����������
��4����2��ʵ���������Һ�����ʵ�����������
| ʵ �� �� �� | 1 | 2 | 3 | 4 | 5 |
| ����ϡ���������/g | 20.0 | 20.0 | 20.0 | 20.0 | 20.0 |
| ʣ����������/g | 6.00 | m | 2.00 | 1.20 | 1.20 |
��l������m��ֵΪ______g��
��2����ʯ��ʯ��Ʒ�Ĵ��ȣ�
��3�����õ�ϡ���������ʵ�����������
��4����2��ʵ���������Һ�����ʵ�����������
| ʵ �� �� �� | 1 | 2 | 3 | 4 | 5 |
| ����ϡ���������/g | 20.0 | 20.0 | 20.0 | 20.0 | 20.0 |
| ʣ����������/g | 6.00 | m | 2.00 | 1.20 | 1.20 |
��l������m��ֵΪ______g��
��2����ʯ��ʯ��Ʒ�Ĵ��ȣ�
��3�����õ�ϡ���������ʵ�����������
��4����2��ʵ���������Һ�����ʵ�����������
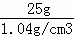 ��25mL����Ҫˮ�����ԼΪ25mL��������Һ��Ҫ�IJ�������������Ͳ����ͷ�ιܣ�
��25mL����Ҫˮ�����ԼΪ25mL��������Һ��Ҫ�IJ�������������Ͳ����ͷ�ιܣ�