��Ŀ����
��1���±���KNO3�ڲ�ͬ�¶�ʱ���ܽ�ȣ���ش��������⣮| �¶ȡ� | 20 | 40 | 60 | 80 | 100 | |
| �ܽ��/g | 13.3 | 31.6 | 63.9 | 110 | 169 | 246 |
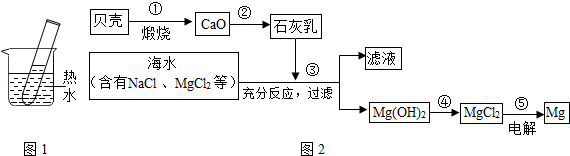
��20��ʱ����װ��20gKNO3������Թ��У�����50gˮ���������ʱ������Һ����Ϊ______g���ٽ����Թܷ���ʢ����ˮ���ձ��У�ͼ1����ʹ�Թ����¶�����60�棬��ʱ�Թ��ڵ��������ܼ�����������______��
������һ����������������KNO3��Һʱ���ܽ����������Ҫ�IJ����������ձ���______��
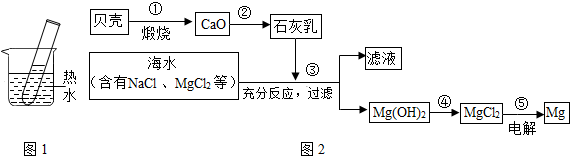
��2��������һ�������Դ���⣬��ˮ���д����������õĻ�ѧ��Դ�������Ȼ�þ���Ȼ��ơ��廯�Ƶȣ�ͼ2Ϊ�ۺ����ú�ˮ�Ʊ�����þ�����̣�����ͼ2��ʾ��ת�������У��������ķ�Ӧ���ڷֽⷴӦ����______���Ӧ˳���ţ���д�����̢ڷ����Ļ�ѧ��Ӧ����ʽ______��

���𰸡���������1���ٸ���KNO3�ڲ�ͬ�¶�ʱ���ܽ�ȣ�����KNO3���ܽ�����¶ȱ仯�Ĺ��ɣ�
�ڸ���20��ʱ��KNO3���ܽ�Ƚ��з������㣻
�۸���������Һ�Ĺ��̷����õ��IJ���������
��2�����ݷ�Ӧ�Ĺ��̷��������ķ�Ӧ���жϷ�Ӧ�����ͣ�д�����Ϸ�Ӧ�ķ���ʽ��
����⣺��1������KNO3�ڲ�ͬ�¶�ʱ���ܽ�ȿ�֪��KNO3���ܽ�����¶ȱ仯�Ĺ��������¶ȵ����߶�����
����20��ʱ��KNO3���ܽ����31.6g�����ܽ�ȵĺ����֪����װ��20gKNO3������Թ��У�����50gˮ�������ֻ���ܽ�15.8g����ʱ������Һ����Ϊ65.8g��������60��ʱ��KNO3���ܽ����110g���ٽ����Թܷ���ʢ����ˮ���ձ��У�ͼ1����ʹ�Թ����¶�����60�棬KNO3ȫ���ܽ⣬��ʱ�Թ��ڵ��������ܼ�����������2��5��
������һ����������������KNO3��Һʱ���ܽ����������Ҫ�IJ����������ձ��Ͳ�������
��2��̼��Ƹ������������ƺͶ�����̼�����ڷֽⷴӦ����������ˮ������������������ڻ��Ϸ�Ӧ��������þ�����ᷴӦ�����Ȼ�þ��ˮ�Ǹ��ֽⷴӦ���Ȼ�þ�ֽ�����þ������Ҳ���ڷֽⷴӦ�����ԣ����ڷֽⷴӦ���Ǣ٢ݣ�
�ڷ����Ļ�ѧ��Ӧ����ʽ�ǣ�CaO+H2O=Ca��OH��2��
�ʴ�Ϊ����1�������¶ȵ����߶�����65.8����2��5����2���٢ݣ�CaO+H2O=Ca��OH��2��
���������⿼����ѧ�����ܽ�ȵ������Ӧ�ã��������˸����ܽ�ȵ����ݷ����ͽ������������������˶��ܽ�ȵ���������⣮
�ڸ���20��ʱ��KNO3���ܽ�Ƚ��з������㣻
�۸���������Һ�Ĺ��̷����õ��IJ���������
��2�����ݷ�Ӧ�Ĺ��̷��������ķ�Ӧ���жϷ�Ӧ�����ͣ�д�����Ϸ�Ӧ�ķ���ʽ��
����⣺��1������KNO3�ڲ�ͬ�¶�ʱ���ܽ�ȿ�֪��KNO3���ܽ�����¶ȱ仯�Ĺ��������¶ȵ����߶�����
����20��ʱ��KNO3���ܽ����31.6g�����ܽ�ȵĺ����֪����װ��20gKNO3������Թ��У�����50gˮ�������ֻ���ܽ�15.8g����ʱ������Һ����Ϊ65.8g��������60��ʱ��KNO3���ܽ����110g���ٽ����Թܷ���ʢ����ˮ���ձ��У�ͼ1����ʹ�Թ����¶�����60�棬KNO3ȫ���ܽ⣬��ʱ�Թ��ڵ��������ܼ�����������2��5��
������һ����������������KNO3��Һʱ���ܽ����������Ҫ�IJ����������ձ��Ͳ�������
��2��̼��Ƹ������������ƺͶ�����̼�����ڷֽⷴӦ����������ˮ������������������ڻ��Ϸ�Ӧ��������þ�����ᷴӦ�����Ȼ�þ��ˮ�Ǹ��ֽⷴӦ���Ȼ�þ�ֽ�����þ������Ҳ���ڷֽⷴӦ�����ԣ����ڷֽⷴӦ���Ǣ٢ݣ�
�ڷ����Ļ�ѧ��Ӧ����ʽ�ǣ�CaO+H2O=Ca��OH��2��
�ʴ�Ϊ����1�������¶ȵ����߶�����65.8����2��5����2���٢ݣ�CaO+H2O=Ca��OH��2��
���������⿼����ѧ�����ܽ�ȵ������Ӧ�ã��������˸����ܽ�ȵ����ݷ����ͽ������������������˶��ܽ�ȵ���������⣮

��ϰ��ϵ�д�
�����Ŀ
��1���±���KNO3�ڲ�ͬ�¶�ʱ���ܽ�ȣ���ش��������⣮
| �¶ȡ� | 0 | 20 | 40 | 60 | 80 | 100 |
| �ܽ��/g | 13.3 | 31.6 | 63.9 | 110 | 169 | 246 |
�ٷ����������ݣ���֪KNO3���ܽ�����¶ȱ仯�Ĺ�����______��
��20��ʱ����װ��20gKNO3������Թ��У�����50gˮ���������ʱ������Һ����Ϊ______g���ٽ����Թܷ���ʢ����ˮ���ձ��У�ͼ1����ʹ�Թ����¶�����60�棬��ʱ�Թ��ڵ��������ܼ�����������______��
������һ����������������KNO3��Һʱ���ܽ����������Ҫ�IJ����������ձ���______��
��2��������һ�������Դ���⣬��ˮ���д����������õĻ�ѧ��Դ�������Ȼ�þ���Ȼ��ơ��廯�Ƶȣ�ͼ2Ϊ�ۺ����ú�ˮ�Ʊ�����þ�����̣�����ͼ2��ʾ��ת�������У��������ķ�Ӧ���ڷֽⷴӦ����______���Ӧ˳���ţ���д�����̢ڷ����Ļ�ѧ��Ӧ����ʽ______��