��Ŀ����
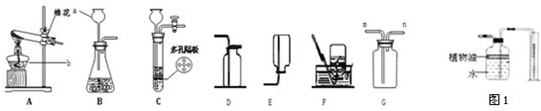
���ͼʾʵ��װ�ã��ش��������⣮

��1��ͼ��a��b���������ƣ�a________��b________��
��2���ø�����ع�����������ѡ�õķ���װ����________������Bװ����ȡ������̼�������Ļ�ѧ��Ӧ����ʽΪ________����Ҫ���Ʒ�Ӧ�����ʣ�Ӧ������©����Ϊ________��A��B������ȡװ�ã���װ��ҩƷǰ��Ӧ��________��
��3��ʵ������ȡ����ʱ������Eװ���ռ�H2��������Ӧ��________��ͨ�루�c����d������
��4��Bװ����������ȡ�������ø��������������ȣ��ŵ���________�����ţ���ͬ����
��������ֻ���������ڷ�Ӧ������ȣ��۷�Ӧ���ڿ��ƣ��ܶԻ�����ȾС��
�⣺��1����Ϥ�����������˽����ǵ����ƣ�ͼ��a��ƿb����ƿ��
��2��ʵ�����ÿ��������ȡ��������Ҫ�þƾ��Ƽ��ȣ���Ҫ��װ��A����Bװ����ȡ������̼ʱ��Ҫ��ϡ���������ʯ����Ҫ�ɷ�CaCO3����Ӧ����ѧ��Ӧʽ�ǣ�CaCO3+2HCl=CaCl2+H2O+CO2������Ҫ���Ʒ�Ӧ�����ʣ�Ӧ������©����Ϊ��Һ©������Ϊ��Һ©���з��ſ��ƣ�����ʹ��Ӧ�濪��ͣ�����Ʒ�Ӧ�����ʣ�Ϊ������˳�����У�������֮ǰҪ���װ�õ������ԣ��ʴ�Ϊ��A��CaCO3+2HCl=CaCl2+H2O+CO2������Һ©��������װ�õ������ԣ�
��3������Eװ���ռ�H2����Ϊ�������ܶ�С�ڿ������ܶȣ�����Ӧ��d�˽��룮�ʴ��ǣ�d��
��4��Bװ����������ȡ�������ø��������������ȣ�Bװ�ò�����ȣ�ʹ�������������У���ѡ�ڣ�
��������1����Ϥ�����������˽����ǵ����ƣ�
��2���ø�����ع�������������Ҫ���ȣ����Ծݴ���������װ�õ�ѡ��ʵ���ҿ��������ʯ��ʯ�ڳ����·�Ӧ��ȡ������̼�����ǿ��Ծݴ�д����ѧ��Ӧʽ�������Һ©���ɿ��Ʒ�Ӧ�����ʣ��ڽ�������ǰһ��Ҫ���װ�õ������ԣ�
��3���������ȿ������ܶ�С��Ӧ��d�˽��������ȸ���ƿ�ڣ�ʹƿ�ڵĿ�����c���ų���
��4�����˽��ø��������ȡ����������Aװ�ã�Ȼ��۲�ͼ�ο�֪��A��Ҫ���ȣ�B������ȣ�Ȼ�����ѡ��ѡ�ɣ�
������������Ҫ��������װ�õ�ѡȡ������ʽ����д�Լ������е�������⣮��Ҫͬѧ�ǶԳ���������װ�ü�������������˽⣮
��2��ʵ�����ÿ��������ȡ��������Ҫ�þƾ��Ƽ��ȣ���Ҫ��װ��A����Bװ����ȡ������̼ʱ��Ҫ��ϡ���������ʯ����Ҫ�ɷ�CaCO3����Ӧ����ѧ��Ӧʽ�ǣ�CaCO3+2HCl=CaCl2+H2O+CO2������Ҫ���Ʒ�Ӧ�����ʣ�Ӧ������©����Ϊ��Һ©������Ϊ��Һ©���з��ſ��ƣ�����ʹ��Ӧ�濪��ͣ�����Ʒ�Ӧ�����ʣ�Ϊ������˳�����У�������֮ǰҪ���װ�õ������ԣ��ʴ�Ϊ��A��CaCO3+2HCl=CaCl2+H2O+CO2������Һ©��������װ�õ������ԣ�
��3������Eװ���ռ�H2����Ϊ�������ܶ�С�ڿ������ܶȣ�����Ӧ��d�˽��룮�ʴ��ǣ�d��
��4��Bװ����������ȡ�������ø��������������ȣ�Bװ�ò�����ȣ�ʹ�������������У���ѡ�ڣ�
��������1����Ϥ�����������˽����ǵ����ƣ�
��2���ø�����ع�������������Ҫ���ȣ����Ծݴ���������װ�õ�ѡ��ʵ���ҿ��������ʯ��ʯ�ڳ����·�Ӧ��ȡ������̼�����ǿ��Ծݴ�д����ѧ��Ӧʽ�������Һ©���ɿ��Ʒ�Ӧ�����ʣ��ڽ�������ǰһ��Ҫ���װ�õ������ԣ�
��3���������ȿ������ܶ�С��Ӧ��d�˽��������ȸ���ƿ�ڣ�ʹƿ�ڵĿ�����c���ų���
��4�����˽��ø��������ȡ����������Aװ�ã�Ȼ��۲�ͼ�ο�֪��A��Ҫ���ȣ�B������ȣ�Ȼ�����ѡ��ѡ�ɣ�
������������Ҫ��������װ�õ�ѡȡ������ʽ����д�Լ������е�������⣮��Ҫͬѧ�ǶԳ���������װ�ü�������������˽⣮

��ϰ��ϵ�д�
�����Ŀ