��Ŀ����
ʵ��������ȡ����. �����Ͷ�����̼���밴Ҫ��ش��й����⡣
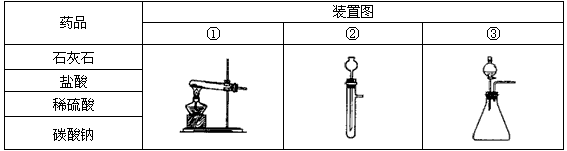
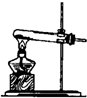
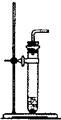
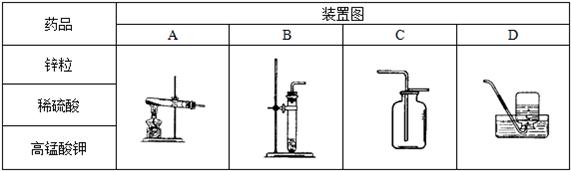
��1�������±��е�ҩƷ��װ�ã�����ȡ���ռ�������ѡ���ҩƷ�� ����Ӧ��װ���� ��
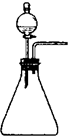
��2����֪��Ȳ��C2H2���������ˮ��CaC2��Ӧ��ȡ������ȡ���������÷���װ���� ���������ȡ��Ȳ��C2H2������Ļ�ѧ��Ӧ����ʽ��CaC2+ === + C2H2����
��1�������±��е�ҩƷ��װ�ã�����ȡ���ռ�������ѡ���ҩƷ�� ����Ӧ��װ���� ��
��2����֪��Ȳ��C2H2���������ˮ��CaC2��Ӧ��ȡ������ȡ���������÷���װ���� ���������ȡ��Ȳ��C2H2������Ļ�ѧ��Ӧ����ʽ��CaC2+ === + C2H2����

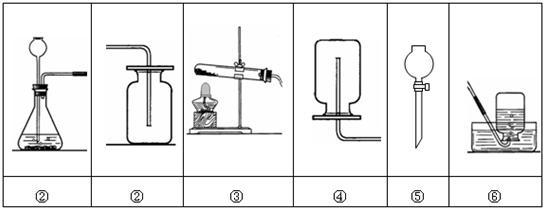
��3���ֽ����±��е�ҩƷ��װ�ã��Ƶ�ֻ�ܺ�������ˮ�Ķ�����̼���壬ѡ���ҩƷ����� ����Ӧ��װ���� ��
��1��������أ�AC��AD
��2��B��2H2O��Ca(OH)2
��3��̼���ƺ�ϡ�����
��2��B��2H2O��Ca(OH)2
��3��̼���ƺ�ϡ�����

��ϰ��ϵ�д�
�����Ŀ

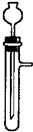

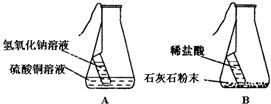
 ��ѧʵ����ѧϰ��ѧ�ͽ��п�ѧ�о�����Ҫ������;����ѧ���������ʵ��Ʊ��������о����塢��������Ļ����������ͼ�ش��й����⣺
��ѧʵ����ѧϰ��ѧ�ͽ��п�ѧ�о�����Ҫ������;����ѧ���������ʵ��Ʊ��������о����塢��������Ļ����������ͼ�ش��й����⣺




