��Ŀ����
����A��F�dz��л�ѧ�е�����ʵ��װ�ã��밴Ҫ����գ�
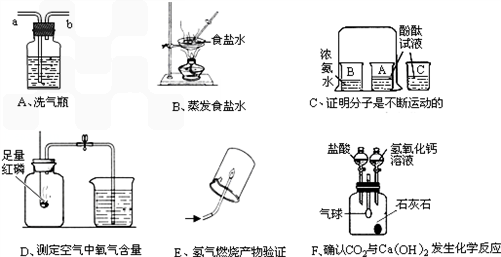
��1��ҽԺ����Aװ�����۲�������������������____���a����b���� Ӧ���Ӳ������������ܽ��ܣ�
��2��Bʵ���в�������������_________________________��
��3��Cʵ�����ձ�C��������________________________��
��4��Dʵ��˵�����������Լռ������______�����в���������______��
��ʵ��ǰ���װ��������
��ʵ��ǰδ�н�ֹˮ��
����ȴ�����º��ٴ�ֹˮ��
�ܺ�������
��5��Eʵ�������Ϊ_______________�������ķ�Ӧ����ʽΪ____________���û�ѧʽ��ʾ����
��6��Fʵ���У��Ƚ�������뼯��ƿ���ٽ�����������Һ���뼯��ƿ��������ı仯�������_____��______������д���������С�����䡱��
��2��Bʵ���в�������������_________________________��
��3��Cʵ�����ձ�C��������________________________��
��4��Dʵ��˵�����������Լռ������______�����в���������______��
��ʵ��ǰ���װ��������
��ʵ��ǰδ�н�ֹˮ��
����ȴ�����º��ٴ�ֹˮ��
�ܺ�������
��5��Eʵ�������Ϊ_______________�������ķ�Ӧ����ʽΪ____________���û�ѧʽ��ʾ����
��6��Fʵ���У��Ƚ�������뼯��ƿ���ٽ�����������Һ���뼯��ƿ��������ı仯�������_____��______������д���������С�����䡱��
��1��b
��2�����裬��ֹ�ֲ��¶ȹ������Һ�ηɽ�
��3����A���Ա�
��4�� �� ��
�� ��
��5����������ɫ���棬�ձ��ڱ���ˮ���� H2+O2 H2O
H2O
��6���������
��2�����裬��ֹ�ֲ��¶ȹ������Һ�ηɽ�
��3����A���Ա�
��4��
 �� ��
�� ����5����������ɫ���棬�ձ��ڱ���ˮ���� H2+O2
 H2O
H2O ��6���������

��ϰ��ϵ�д�
 �����ߴ���ϵ�д�
�����ߴ���ϵ�д�
�����Ŀ





