��Ŀ����
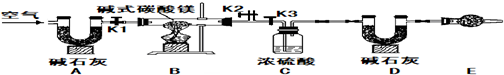
����ɫ��ĩA����ɫҺ��B��ϣ�����һ����ɫ��ζ������C�����Ȱ���ɫ����E����ͬʱ�õ�A��C����ɫ����C��ʹ�����ǵ�ľ����ȼ����������ɫ����D����ʢ��C�ļ���ƿ��ȼ�գ��������䣬����һ�ֺ�ɫ����F�����ų����������ƶ�A��B��C��D��E��F����ʲô���ʣ��ѧ���ţ���
��1��A______��B______��C______��D______��E______��F______��
��2��D��ʢ��C�ļ���ƿ��ȼ�գ�����ƿ�ױ����һЩ______��Ŀ����______��
��1��A______��B______��C______��D______��E______��F______��
��2��D��ʢ��C�ļ���ƿ��ȼ�գ�����ƿ�ױ����һЩ______��Ŀ����______��
������ʱ��һ��ץסʵ�鷴Ӧ������Ĺؼ��ģ�
��1�����ȴ�������ʹ�����ǵ�ľ����ȼ������C����ȷ��CΪ����������D����ȼ�գ��������䣬���ɺ�ɫ����F�����п����ж�DΪ������FΪ����������������C��������֪��Һ��A�ǹ������⣬B�Ƕ������̣������Ϻ�ɫ����E���Եõ��������̺���������E�Ǹ�����أ�
��2����˿��������ȼ�գ�ƿ��Ҫ������ϸɳ��ʢ����ˮ����ֹ��ֹ�����ヲ��ը��ƿ�ף�
�ʴ�Ϊ����1��MnO2��H2O2��O2 ��Fe��KMnO4��Fe3O4��
��2����һЩˮ��ɳ����ֹ�����ヲ��ը��ƿ�ף�
��1�����ȴ�������ʹ�����ǵ�ľ����ȼ������C����ȷ��CΪ����������D����ȼ�գ��������䣬���ɺ�ɫ����F�����п����ж�DΪ������FΪ����������������C��������֪��Һ��A�ǹ������⣬B�Ƕ������̣������Ϻ�ɫ����E���Եõ��������̺���������E�Ǹ�����أ�
��2����˿��������ȼ�գ�ƿ��Ҫ������ϸɳ��ʢ����ˮ����ֹ��ֹ�����ヲ��ը��ƿ�ף�
�ʴ�Ϊ����1��MnO2��H2O2��O2 ��Fe��KMnO4��Fe3O4��
��2����һЩˮ��ɳ����ֹ�����ヲ��ը��ƿ�ף�

��ϰ��ϵ�д�
 ��������ܸ�ϰϵ�д�
��������ܸ�ϰϵ�д�
�����Ŀ

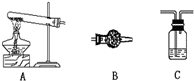


 ��ʵ��̽�����������¾���һֱ�����ǵ���Ըͬѧ�Ƕ��Խ�����ܽᡢ��ʵ�飬����һ����ͼ����װ�ã�����С�����̽������B��ʵ��ҩƷ������Ҫ�Լ�ѡ��
��ʵ��̽�����������¾���һֱ�����ǵ���Ըͬѧ�Ƕ��Խ�����ܽᡢ��ʵ�飬����һ����ͼ����װ�ã�����С�����̽������B��ʵ��ҩƷ������Ҫ�Լ�ѡ��