摘要:A.N2(g)+3H2(g) = 2NH3kJ?mol-1 B.N2(g)+3H2(g) = 2NH3kJ?mol-1
网址:http://m.1010jiajiao.com/timu_id_161412[举报]
化学反应N2+3H2===2NH3的能量变化如右图所示,该反应的热化学方程式是
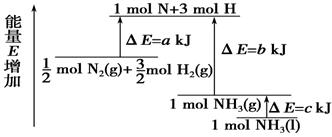
A.N2(g)+3H2(g)===2NH3(l) ΔH=2(a-b-c)kJ·mol-
B.N2(g)+3H2(g)===2NH3(g) ΔH=2(b-a)kJ·mol-1
C. 1/2 N2 (g)+3/2H2(g)===NH3(l) ΔH=(b+c-a)kJ·mol-1
D. 1/2 N2(g)+3/2H2(g)===NH3(g) ΔH=(a+b)kJ·mol-1
查看习题详情和答案>>
下列反应中符合图象的是
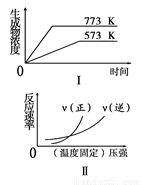
A.N2(g)+3H2(g) 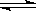 2NH3(g) ΔH<0
2NH3(g) ΔH<0
B.2SO3(
g) 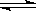 2SO2(g)+O2(g) ΔH>0
2SO2(g)+O2(g) ΔH>0
C.4NH3(g)+5O2(g)  4NO(g)+6H2O(g)
ΔH<0
4NO(g)+6H2O(g)
ΔH<0
D.H2(g)+CO(g)  C(s)+H2O(g) ΔH>0
C(s)+H2O(g) ΔH>0
查看习题详情和答案>>
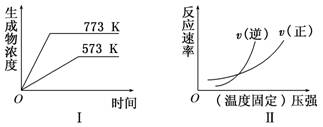
化学反应N2+3H2=2NH3的能量变化如图所示,E是正值,该反应的热化学方程式是( )
| A.N2(g)+3H2(g)=2NH3(1); △H=2(a-b-c)kJ·mol-1 |
| B.N2(g)+3H2(g)=2NH3(g);△H=2(b-a)kJ·mol-1 |
C. N2(g)+ N2(g)+ H2(g)=NH3(1);△H=(b+c-a)kJ·mol-1 H2(g)=NH3(1);△H=(b+c-a)kJ·mol-1 |
D. N2(g)+ N2(g)+ H2(g)=NH3(g); △H=(a+b)kJ·mol H2(g)=NH3(g); △H=(a+b)kJ·mol |
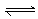 2NH3(g) ΔH<0
2NH3(g) ΔH<0
 C、N2(g)+H2(g) = NH3(l) ⊿H = (b+c—a)kJ/mol
C、N2(g)+H2(g) = NH3(l) ⊿H = (b+c—a)kJ/mol  N2(g)+
H2(g) =NH3(g)
⊿H = (a+b)kJ/mol [来源:Z.xx.k.Com][来源:ZXXK]
N2(g)+
H2(g) =NH3(g)
⊿H = (a+b)kJ/mol [来源:Z.xx.k.Com][来源:ZXXK]