��Ŀ����
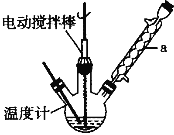
����Ŀ��ij�о���ѧϰС���ͬѧ��������װ�ý����Ҵ���������ʵ�顣������ش�
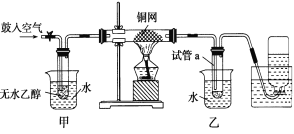
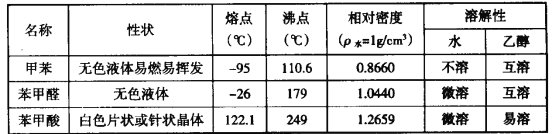
��1����д��ʵ������е��ܵĻ�ѧ��Ӧ����ʽ____________________________________��
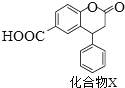
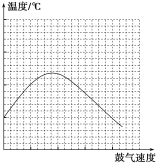
��2���ڲ��Ϲ������������£���ȥ�ƾ��ƣ���Ӧ���ܼ������У�˵���Ҵ��Ĵ�������Ӧ��________��Ӧ����һ���о������������ٶ��뷴Ӧ��ϵ���¶ȹ�ϵ��������ͼ��ʾ�������ٶȹ��죬��Ӧ��ϵ�¶ȷ����½���ԭ��___________________________��
��3����Ӧ����һ��ʱ������Թ�a�����ռ�����ͬ�����ʣ�������_________________������ƿ���ռ������������Ҫ�ɷ���_______________��
��4���ڼ���������Ĺ����У�һλͬѧ��żȻ�ط�������ˮ�м�����ȩ��Һ����ˮ��ɫ�������ˮ��ɫ��ԭ��С���ͬѧ�Ǿ���������������ֲ��룺
����ˮ����ȩ����Ϊ�������ˮ����ȩ�����ӳɷ�Ӧ��
�������һ����ʵ�飬̽����һ�ֲ�����ȷ��________________________________��
���𰸡���1��2CH3CH2OH��O2![]() 2CH3CHO��2H2O
2CH3CHO��2H2O
��2�����ȣ� ���������彫��Ӧ��ϵ�е���������
��3����ȩ���Ҵ���ˮ������
��4����pH��ֽ�����ˮ����ɫ����Һ������ԡ�������������ǿ����������ȷ�����������Լ�������������ȷ������������Ҳ����
��������
�����������1���Ҵ���ͨ������ʱ�����Ա���������Ϊ��ȩ����Ӧ�ķ���ʽΪ��2CH3CH2OH+O2![]() 2CH3CHO+2H2O��
2CH3CHO+2H2O��
�ʴ�Ϊ��2CH3CH2OH+O2![]() 2CH3CHO+2H2O��
2CH3CHO+2H2O��
��2��ʵ��ʱ���ڲ��Ϲ������������£���ȥ�ƾ��ƣ���Ӧ���ܼ������У�˵����Ӧ����������ȼ��ɽ��е��ף�˵���÷�Ӧ�Ƿ��ȵķ�Ӧ����Ӧ�ų����ȵĶ��ٺ��Ҵ��Լ����������йأ������෴Ӧ���ȶ࣬���Կ�ʼ���¶����ߣ����ǹ���������Ὣ��ϵ�е��������ߣ����Ժ���¶Ƚ�����
�ʴ�Ϊ�����ȣ����������彫��Ӧ��ϵ�е��������ߣ�
��3���Ҵ��Ĵ�����ʵ���е����ʣ��Ҵ�����ȩ��ˮ�ķе�ߵͲ�ͬ�����Թ�a�����ռ���Щ��ͬ�����ʣ������ijɷ���Ҫ�ǵ����������������μӷ�Ӧ��ʣ�����Ҫ�ǵ�����
�ʴ�Ϊ����ȩ���Ҵ���ˮ��������
��5��������ȩ������������������廯�����ɣ���Һ��ǿ���ԣ����Ƿ����ӳɷ�Ӧ�������ɱ��ͺ����л����Һ��Ϊ���ԣ�������pH��ֽ�����ˮ����ɫ����Һ������ԣ�������������ǿ����������ȷ�����������Լ�������������ȷ��
�ʴ�Ϊ����pH��ֽ�����ˮ����ɫ����Һ������ԣ�������������ǿ����������ȷ�����������Լ�������������ȷ��

 һ����ʦ����Ӧ����������һ��ȫϵ�д�
һ����ʦ����Ӧ����������һ��ȫϵ�д� Сѧѧϰ�ð���ϵ�д�
Сѧѧϰ�ð���ϵ�д�