��Ŀ����
��Ϥ��ʹ����ѧ��ѧʵ���г�����������;���ǻ�ѧѧϰ�Ļ���Ҫ���Իش��������⡣
(1)������a.��Һ©����b���Լ�ƿ��c������ƿ��d���ζ��ܡ�e������ƿ��f����Ͳ��g��������ƽ�У����С�0���̶ȵ���________(�����)��
(2)��ĥɰ�������Ӳ��������ܷ��Ե�һ�ִ������գ������������У�û���õ���ĥɰ�����մ�������________(�����)��
a���Թܡ� b����Һ©���� c�����ιܵ��Լ�ƿ(��ƿ)
d������ƿ��e����ʽ�ζ��ܡ�f����ʽ�ζ���
(3)��װ�üס��ҡ������齺�����һ��װ��(�������Ѽ��)����������ȡ���ռ�NH3��HCl���壬�ɹ�ѡ���Һ���Լ��У�Ũ���ᡢŨ���ᡢŨ��ˮ�������Լ�Ϊ��ɫʯ����Һ���Իش��������⣺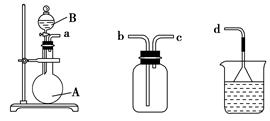
�������ס��� �����ҡ����� ����
������ȡij����Ĺ����У����е�ʯ����Һ��죬����ƿ�е��Լ�A���Һ©���е��Լ�B��Ϊ��ɫҺ�壬���Լ�AΪ________����ȡ������������B��������________��________��
ͨ��������ɫʯ����Һ���������˵�����������Ѽ�������ʯ����Һ����������ƿ����Ӧ����Ĺ����Լ�AΪ________��
�����������ռ���������������װ�õ�˳���ǣ�a�D��____�D��_____�D��d(��ӿڴ���)��
��(1)dg��(2)af��(3)��Ũ���ᡡ��ˮ�ԡ�����ˮ�ų������ȡ�NaOH��CaO���ʯ�ҡ���c��b
����

�����й�ʵ�������ȷ����
| A���ù����Ȼ�狀�����������ȡ�������������Թܴ�����̨��ȡ����������ϴ�� |
| B���ñ���ȡ��ˮ�е���ʱ������ı���Һ�ӷ�Һ©���¿ڷų� |
| C���������Ƶ�����ʵ��ʱ��ʣ����ƷŻ�ԭ�Լ�ƿ |
| D������ڵ�NaOHϡ��Һ�е���FeCl3������Һ�����Ʊ�Fe��OH��3���� |
�����������ʵķ�����ȷ����(����)
| A���������������ڶ���̼�� |
| B��ˮ���������ڴ��������IJ���ƿ�� |
| C�������������ھƾ��� |
| D��������������ú���� |
�����йػ�ѧʵ���˵������ȷ����
| A���ձ����������Թܡ���ƿ�������þƾ���ֱ�Ӽ��� |
| B��������Ӧ����ȩ������Cu(OH)2��Ӧ��ʵ������ȡ��ϩ��������ˮԡ���� |
| C����ȡ����������ʱ�������������ſ������ռ� |
| D��ʯ�͵ķ���ʵ������ȡ��Ȳ����ȡ����ˮ��Ҫ�õ�����װ�� |
��6�֣���ͼ��A��B��C��D�dz�������ͨ�����Լ�ƿ����������Լ��������д���ʺ�ʢ�ŵ��Լ�ƿ����������ڣ�
a��Ũ���� b��̼������Һ c����Ƭ d��Ũ���� e������������Һ f����������
| A | B | C | D |
 |  |  |  |
| �� �� | �� �� | �� �� | �� �� |
����ҩƷ��װ�ú������������Ӧʵ�����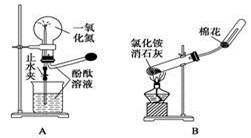
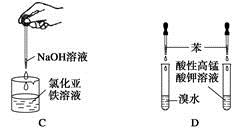
| A����Ȫʵ�� |
| B��ʵ������ȡ���ռ����� |
| C���Ʊ����������� |
| D����֤�����Ƿ���̼̼˫�� |
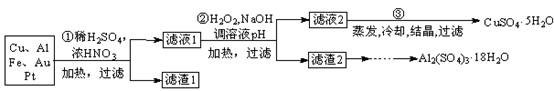
 12Na2CrO4+3Fe2O3+7KCl+12H2O
12Na2CrO4+3Fe2O3+7KCl+12H2O