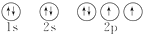��Ŀ����
9��Ŀǰ�������嵥�ʵķ�����Ҫ��ˮ����������ȡ���ȣ�ij��ȤС��ͨ���������������������·����Ӹ������Һ������0.27%���л����ӷ���Br2��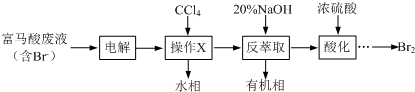
��1������X����Ҫ����Ҫ��������Ϊ��Һ©��������ȡʱ����20%��NaOH��Һ�������ӷ���ʽΪBr2+2OH-=Br-+BrO-+H2O��
��2������ȡ����ˮ���ữʱ���軺������Ũ���ᣬ�����ñ�ˮԡ��ȴ��ԭ���ǣ�����Br2�Ļӷ���
��3����Ĵ�ͳ��������Ϊ�Ȳ����������������ÿ���ˮ������Br2�������봫ͳ������ȣ���ȡ�����ŵ���û�в����ж�����Cl2����������
��4���ҹ���ˮ�����ŷű��涨����ˮ�б��ӵĺ������ó���1.00mg/L��ʵ���ҿ���һ��Ũ�ȵ���ˮ�ⶨij��ˮ�б��ӵĺ�������ԭ�����£�

���������Ӧ��ʵ�鲽�裺
����1��ȷ��ȡ25.00mL�����ˮ��250mL��ƿ�У�
����2����4.5mL 0.02mol/L��ˮѸ�ټ��뵽��ƿ�У�����ƿ������
����3����ƿ��������ƿ�м��������0.1mol/L KI��Һ����
����4������2��3�ε�����Һ������0.01mol/L Na2S2O3����Һ�ζ����յ㣬���� Na2S2O3��Һ15mL������Ӧԭ����I2+2Na2S2O3�T2NaI+Na2S4O6��
����5����ʵ�鲽��1��4�ظ�2�Σ�
�ڸ÷�ˮ�б��ӵĺ���Ϊ18.8mg/L��
�۲���3������ʱ��ϳ������õķ�ˮ�б��ӵĺ���ƫ�ͣ�
���ƫ�ߡ�����ƫ�͡�����Ӱ�족����
���� �������Һ�к���Br-����������嵥�ʣ��������Ȼ�̼��ȡ���Һ���ټ����������Ʒ���ȡ�õ��������ߺʹ���������ӵ���Һ�����������ữ����������ԭ��Ӧ�����嵥�ʣ�
��1������XΪ��ȡ��Һ����Ҫ�ձ�����Һ©��������������ȡʱ����20%��NaOH��Һ��Ӧ�����廯�ơ��������ƺ�ˮ��
��2���嵥���ӷ���Ũ�����ܽ���ȷ�����
��3�����ж����������μӷ�Ӧ���������̻�����
��4���������������Һ�ζ��ⵥ�ʣ����������Һ��ָʾ����
�����ݻ�ѧ����ʽ������ϵ���㣻
�۲���3������ʱ�䳤���µ���KI��������
��� �⣺�������Һ�к���Br-����������嵥�ʣ��������Ȼ�̼��ȡ���Һ���ټ����������Ʒ���ȡ�õ��������ߺʹ���������ӵ���Һ�����������ữ����������ԭ��Ӧ�����嵥�ʣ�
��1������XΪ��ȡ��Һ����Ҫ�ձ�����Һ©��������������ȡʱ����20%��NaOH��Һ��Ӧ�����廯�ơ��������ƺ�ˮ����Ӧ�����ӷ���ʽΪ��Br2+2OH-=Br-+BrO-+H2O��
�ʴ�Ϊ����Һ©����Br2+2OH-=Br-+BrO-+H2O��
��2���嵥���ӷ���Ũ�����ܽ���ȷ���������ȡ����ˮ���ữʱ���軺������Ũ���ᣬ�����ñ�ˮԡ��ȴ��ԭ���Ǽ����嵥�ʵĻӷ���
�ʴ�Ϊ������Br2�Ļӷ���
��3�����ж����������μӷ�Ӧ���������̻�����
�ʴ�Ϊ��û�в����ж�����Cl2����������
��4���������������Һ�ζ��ⵥ�ʣ����������Һ��ָʾ��������2��3�ε�����Һ������0.01mol/L Na2S2O3����Һ�ζ����յ㣻
�ʴ�Ϊ������2��3�ε�����Һ��
�����ݻ�ѧ����ʽ������ϵ���㣬��4.5mL 0.02mol/L��ˮѸ�ټ��뵽��ƿ�У�n��Br2��=0.0045L��0.02mol/L=0.00009mol�����к͵⻯�ط�Ӧ���嵥�����ʵ���
2KI��Br2��I2��2Na2S2O3����
1 2
n 0.01mol/L��0.015L
n=0.000075mol
�뱽�ӷ�Ӧ���嵥��=0.00009mol-0.000075mol=0.000015mol
�����ʵ���=$\frac{1}{3}$��0.000015mol=0.000005mol
�÷�ˮ�б��ӵĺ���Ϊ=$\frac{1000ml}{25ml}$��0.000005mol��94g/mol=0.0188g/L=18.8mg/L��
�ʴ�Ϊ��18.8��
�۲���3������ʱ�䳤���µ���KI���������ⶨ���������٣��ⶨ���ƫ�ͣ�
�ʴ�Ϊ��ƫ�ͣ�
���� ���⿼���˷�ˮ�����ʺ����IJⶨʵ��̽�����������ʵķ����жϣ�ע��ζ�ʵ��Ĺ��̷���Ӧ�ã����ջ����ǹؼ�����Ŀ�Ѷ��еȣ�

 ��У����ϵ�д�
��У����ϵ�д� ��֪NH3��HCl������������Ȫʵ������壬����ͬ��ͬѹ���õ������ƿһ���ռ���NH3����һ���ռ�HCl��N2�Ļ�����壬��ͼ��ʾ����Ȫʵ��ֹͣ��������ƿ����Һ�����ʵ����ʵ���Ũ�ȣ�c�������ʵ�����������w����ϵ�ǣ�������
��֪NH3��HCl������������Ȫʵ������壬����ͬ��ͬѹ���õ������ƿһ���ռ���NH3����һ���ռ�HCl��N2�Ļ�����壬��ͼ��ʾ����Ȫʵ��ֹͣ��������ƿ����Һ�����ʵ����ʵ���Ũ�ȣ�c�������ʵ�����������w����ϵ�ǣ�������| A�� | c��w����ͬ | B�� | c��w����ͬ | C�� | c��ͬ��w��ͬ | D�� | w��ͬ��c��ͬ |
| A�� | SO2��SiO2��CO������������ | |
| B�� | ˮ�ࡢ�մɡ������ˮ����Ϊ�����β�Ʒ | |
| C�� | �ռ�����ᡢ���Ȼ�̼��Ϊ����� | |
| D�� | Ư�ۡ�ˮ��������ˮ�����;�Ϊ����� |
| A�� | ʵ������H2O2������ | B�� | ��ҵ���ð��������� | ||
| C�� | ʵ������ŨHNO3��NO2���� | D�� | ʵ�������Ȼ���ư��� |
| ѡ�� | ���� | ���� |
| A | ��Ag+���ӵ���Һ�еμ�ϡ����������ɫ���� | �μ�ϡ���������ɫ��������Һ��һ������Ag+���� |
| B | ��������Һ�еμӹ�����ˮ��ȡ������������ | ��������Һ�еμӹ���NaOH��Һ���յò��������������� |
| C | Fe��OH��3�����еμ�ϡ���������Ȼ�����ˮ | Fe��OH��3�����еμ�HI���ɵ⻯����ˮ |
| D | ͭƬ������ϡ���� | ͭƬҲ������ϡ���� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ��Һ�Ե����ԣ�������е�ɣ����Խ�����ȶ� | |
| B�� | ��������������ֽ������������Ĥ�����Կ��������ķ������ƽ��� | |
| C�� | ���ö����ЧӦ������Һ�ͽ��� | |
| D�� | ��Ӿʵ��˵���˽���������������Һ�е������ӻ������Ӷ����е�� |
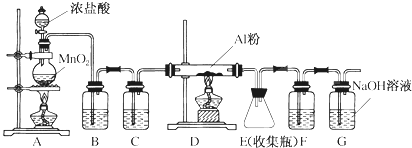
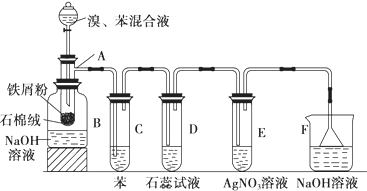
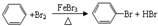 �����л���Ӧ������Ϊȡ����Ӧ��
�����л���Ӧ������Ϊȡ����Ӧ��