��Ŀ����
����Ŀ��I������![]() ��
��![]() ��Һʱ�����ֻ�����������⣬��Һ��Ũ����α仯�����ƫ��ƫС�����䡱��
��Һʱ�����ֻ�����������⣬��Һ��Ũ����α仯�����ƫ��ƫС�����䡱��
��1��������ƿ�м�����ˮ���ڿ̶��ߣ�����_____________��
��2������ʱ�۲�̶������ӣ�����_____________��
II����1��ʵ���Ҽ��ȹ����Ȼ�狀��������ƵĻ������ȡ������Ӧ�Ļ�ѧ����ʽΪ��__________________________��
��2����������װ����ѡ��ʵ�����Ʊ����ķ���װ�ã�_____________���������ĸ����ո���ͬ����Ϊ�˵õ������İ������������Լ���ѡ����ﰱ���Լ���_____________��
װ���У�
A B
B![]() C
C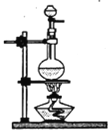 D
D![]() E
E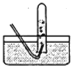 F
F![]() G
G![]() H
H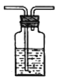
�Լ��У�a��![]() ��Һb������ʯ��ˮc��Ũ����d������
��Һb������ʯ��ˮc��Ũ����d������![]() ��Һe����ʯ��f��Ʒ����Һg��ʪ��ĺ�ɫʯ����ֽ
��Һe����ʯ��f��Ʒ����Һg��ʪ��ĺ�ɫʯ����ֽ
���𰸡�ƫ�� ƫС 2NH4Cl+Ca(OH)2 �� 2NH3��+CaCl2+2H2O A e
��������
����n=cV�ɵã�һ�����ʵ���Ũ����Һ���Ƶ����������ʵ����ʵ���n����Һ�����V����ģ�������һ�����ʵ���Ũ����Һʱ����V������ֵ��ʱ����ʹ������ҺŨ��ƫС����V������ֵСʱ������ʹ������ҺŨ��ƫ��
I����1��������ƿ�м�����ˮ���ڿ̶��ߣ�ˮƫС������ƫ��
��2������ʱ�۲�̶������ӣ���Ұ��ˮλ����̶���ʱ��ʵ�ʼ�ˮ����������ƫС��
II����1��ʵ���Ҽ��ȹ����Ȼ�狀��������ƵĻ������ȡ������Ӧ�Ļ�ѧ����ʽΪ��2NH4Cl+Ca(OH)2 �� 2NH3��+CaCl2+2H2O��
��2��ʵ�����Ʊ����ǹ̹̼�����װ�ã�������װ��ΪA��Ϊ�˵õ������İ�������Ϊ�������壬���ﰱ���Լ�������ˮ�ԣ��Ҳ�����������ʷ�����Ӧ����ѡe��