��Ŀ����
������ˮ��Һ�п��ܴ��ڵ���ƽ�⡢�ε�ˮ��ƽ�������ܽ�ƽ�⣬���Ƕ��ɿ����ǻ�ѧƽ���һ�֡��������ѧ��ѧ֪ʶ�ش��������⣺
��12�֣���֪0.01mol/L��NaHCO3��Һ�ʼ��ԡ�
(1)����Һ�д���������ƽ����ϵ���õ��뷽��ʽ�����ӷ���ʽ��ʾ��
�� �� ��
��2����Һ�й��� �ֲ�ͬ�����ӣ�ָ���Ӻ����ӣ���
��3������Һ�е�����Ũ���ɴ�С��˳���ǣ� ��
��4�������Һ�м���AlCl3��Һ���а�ɫ��״ �������ɣ�ͬʱ����һ����ɫ��ζ����ʹ����ʯ��ˮ����ǵ����壬д���÷�Ӧ�����ӷ���ʽ�� ��
�������ɣ�ͬʱ����һ����ɫ��ζ����ʹ����ʯ��ˮ����ǵ����壬д���÷�Ӧ�����ӷ���ʽ�� ��
��6�֣������ᡢ���ᡢ����������Һ���Իش�
��1����������Һ��Ũ�Ⱦ�Ϊ0.1 mol?L-1����c(H+) ����Ϊa mol?L-1��b mol?L-1��c mol?L-1�����С˳��Ϊ ���к�һ����NaOH��Һ��������ʱ������������������������V1L��V2L��V3L�����С��ϵΪ ��
��2����������Һ��pH��Ϊ2���ֱ�ȡ10mL������Һ��ˮϡ����1000mL,��ʱ������Һ��pH1��pH2��pH3֮��Ĺ�ϵΪ ���á���������������=����ʾ��pH1 �����������ᡢ pH2 �����������ᡢpH3 �����������ᣩ
��4�֣���֪��������AgCl����ı�����Һ�д�������ƽ�⣺AgCl(s) Ag��(aq)��Cl��(aq),����25��ʱ��AgCl��Ksp=1.8��10-10���ֽ�����AgCl����ֱ��������Һ���У���100mL����ˮ
Ag��(aq)��Cl��(aq),����25��ʱ��AgCl��Ksp=1.8��10-10���ֽ�����AgCl����ֱ��������Һ���У���100mL����ˮ
��100mL0.3mol/L AgNO3��Һ ��100mL0.1mol/L MgCl2��Һ��ֽ������ȴ��25��ʱ����ʱc(Ag��)�ɴ�С��˳��Ϊ ������ţ�����Һ����c(Cl-)= ��
��12�֣���֪0.01mol/L��NaHCO3��Һ�ʼ��ԡ�
(1)����Һ�д���������ƽ����ϵ���õ��뷽��ʽ�����ӷ���ʽ��ʾ��
�� �� ��
��2����Һ�й��� �ֲ�ͬ�����ӣ�ָ���Ӻ����ӣ���
��3������Һ�е�����Ũ���ɴ�С��˳���ǣ� ��
��4�������Һ�м���AlCl3��Һ���а�ɫ��״
 �������ɣ�ͬʱ����һ����ɫ��ζ����ʹ����ʯ��ˮ����ǵ����壬д���÷�Ӧ�����ӷ���ʽ�� ��
�������ɣ�ͬʱ����һ����ɫ��ζ����ʹ����ʯ��ˮ����ǵ����壬д���÷�Ӧ�����ӷ���ʽ�� ����6�֣������ᡢ���ᡢ����������Һ���Իش�
��1����������Һ��Ũ�Ⱦ�Ϊ0.1 mol?L-1����c(H+) ����Ϊa mol?L-1��b mol?L-1��c mol?L-1�����С˳��Ϊ ���к�һ����NaOH��Һ��������ʱ������������������������V1L��V2L��V3L�����С��ϵΪ ��
��2����������Һ��pH��Ϊ2���ֱ�ȡ10mL������Һ��ˮϡ����1000mL,��ʱ������Һ��pH1��pH2��pH3֮��Ĺ�ϵΪ ���á���������������=����ʾ��pH1 �����������ᡢ pH2 �����������ᡢpH3 �����������ᣩ
��4�֣���֪��������AgCl����ı�����Һ�д�������ƽ�⣺AgCl(s)
 Ag��(aq)��Cl��(aq),����25��ʱ��AgCl��Ksp=1.8��10-10���ֽ�����AgCl����ֱ��������Һ���У���100mL����ˮ
Ag��(aq)��Cl��(aq),����25��ʱ��AgCl��Ksp=1.8��10-10���ֽ�����AgCl����ֱ��������Һ���У���100mL����ˮ��100mL0.3mol/L AgNO3��Һ ��100mL0.1mol/L MgCl2��Һ��ֽ������ȴ��25��ʱ����ʱc(Ag��)�ɴ�С��˳��Ϊ ������ţ�����Һ����c(Cl-)= ��
��12�֣�(1) �� HCO3�� H���� CO32�� �� HCO3���� H2O
H���� CO32�� �� HCO3���� H2O H2CO3�� OH��
H2CO3�� OH��
�� H2O H���� OH�� �������Ⱥ�˳��
H���� OH�� �������Ⱥ�˳��
��2��7
��3��c(Na��)��c(HCO3��)��c(OH��)��c(H��)��c(CO32��)
��4��3HCO3�� + Al3+ ="==" Al(OH)3�� + 3CO2��
��6�֣���1��b��a��c V2��V1=V3 ��2��pH1=pH2��pH3
��4�֣� �ڢ٢� 6��10-10 mol/L
 H���� CO32�� �� HCO3���� H2O
H���� CO32�� �� HCO3���� H2O H2CO3�� OH��
H2CO3�� OH���� H2O
 H���� OH�� �������Ⱥ�˳��
H���� OH�� �������Ⱥ�˳����2��7
��3��c(Na��)��c(HCO3��)��c(OH��)��c(H��)��c(CO32��)
��4��3HCO3�� + Al3+ ="==" Al(OH)3�� + 3CO2��
��6�֣���1��b��a��c V2��V1=V3 ��2��pH1=pH2��pH3
��4�֣� �ڢ٢� 6��10-10 mol/L
��

��ϰ��ϵ�д�
 ����ѵ�����⿼ϵ�д�
����ѵ�����⿼ϵ�д�
�����Ŀ
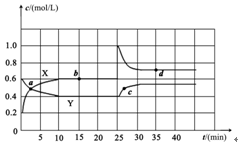
 2B(g)+C(g)����H>0����ƽ��ʱ��ҪʹV���������͡�C(A)����Ӧ��ȡ�Ĵ�ʩ�ǣ� ��
2B(g)+C(g)����H>0����ƽ��ʱ��ҪʹV���������͡�C(A)����Ӧ��ȡ�Ĵ�ʩ�ǣ� �� mZ(g)����H����a kJmol��1(a>0)�����мס������ݻ�����ҹ̶����ܱ��������ڱ��ָ��¶Ⱥ㶨�������£����ܱ���������ͨ��2 mol X��1 mol Y���ﵽƽ��״̬ʱ���ų�����b kJ�����ܱ���������ͨ��1 mol X��0.5 mol Y���ﵽƽ��ʱ���ų�����c kJ����b>2c����a��b��m��ֵ���ϵ��ȷ����
mZ(g)����H����a kJmol��1(a>0)�����мס������ݻ�����ҹ̶����ܱ��������ڱ��ָ��¶Ⱥ㶨�������£����ܱ���������ͨ��2 mol X��1 mol Y���ﵽƽ��״̬ʱ���ų�����b kJ�����ܱ���������ͨ��1 mol X��0.5 mol Y���ﵽƽ��ʱ���ų�����c kJ����b>2c����a��b��m��ֵ���ϵ��ȷ���� 4NO(g) + 6H2O(g)
4NO(g) + 6H2O(g) 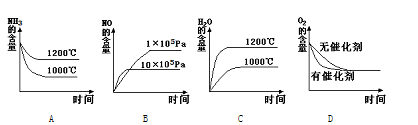
 2Z(g)����H<0���ڲ�ͬ�¶ȣ�T1��T2���£�����Z�����ʵ���n�뷴Ӧʱ��t�Ĺ�ϵ��ͼ��ʾ���������ж�����ȷ���ǣ� ��
2Z(g)����H<0���ڲ�ͬ�¶ȣ�T1��T2���£�����Z�����ʵ���n�뷴Ӧʱ��t�Ĺ�ϵ��ͼ��ʾ���������ж�����ȷ���ǣ� ��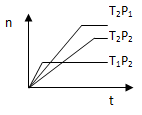
 zC���ﵽƽ�⣬����д���пհף�
zC���ﵽƽ�⣬����д���пհף�