��Ŀ����
A�������ʽṹ�����ʡ�ģ�飨12�֣�
��ï����(C5H5)2 Fe������һ�����������Ӻ�2������ϩ�������ӹ��ɣ����ķ��ֿ���˵���л������������о��о�����̱�������¼����������˽����л��������о��������ٽ��˽����л���ѧ�ķ�չ����ï�������û�ԭ�����뻷���ϩ�ڵ�����Χ�з�����Ӧ���Ƶã�
2C5H6��Fe(��ԭ����)![]() (C5H5)2 Fe��H2��
(C5H5)2 Fe��H2��
��ش��������⣺
��1��д�����������ӵĻ�̬�����Ų�ʽ�� ��
��2����ï�����۵���173�棨��100��ʱ��ʼ���������е���249�棬������ˮ�������ڱ������ѵȷǼ����ܼ����ݴ˿��ƶ϶�ï������Ϊ ���壬�ɴ˿ɽ�һ����֪���ڶ�ï���ṹ�У�C5H5����Fe2+֮���γɵĻ�ѧ�������� ��
��3�������ϩ�ĽṹʽΪ�� ��ͼ�����ֽ�����̼ԭ�ӱ�ţ�������5��̼ԭ���в�ȡsp3�ӻ����� ����д��ţ���
��ͼ�����ֽ�����̼ԭ�ӱ�ţ�������5��̼ԭ���в�ȡsp3�ӻ����� ����д��ţ���![]() �ǻ�ï��ϩ��һ��ͬ���칹�壬������ӽṹ�д���ͬһƽ���ϵ�ԭ�Ӹ�������� ����
�ǻ�ï��ϩ��һ��ͬ���칹�壬������ӽṹ�д���ͬһƽ���ϵ�ԭ�Ӹ�������� ����
��4��ԭ����Ŀ�͵�����������۵�����������ͬ������Ϊ�ȵ����壬�ȵ�����������ƵĽṹ��������N2���ӻ�Ϊ�ȵ�����Ķ����������� ���ѧʽ���������ʽ�� ��
B����ʵ�黯ѧ��ģ�飨12�֣�
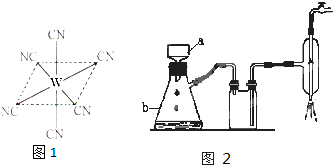
��12�֣�ij����С��Ϊ�˲ⶨij�Ȼ��ȣ�SrCl2����Ʒ�Ĵ��ȣ��ڽ�ʦָ������������·�����
��ȡ1.0g��Ʒ�ܽ�������ˮ�У������м��뺬AgNO32.38g��AgNO3��Һ����Һ�г�Cl�D�⣬����������Ag+��Ӧ���ɳ��������ӣ���Cl�D����ȫ��������Ȼ���ú�Fe3+����Һ��ָʾ������NH4SCN����Һ�ζ�ʣ���AgNO3��ʹʣ���Ag+��AgSCN��ɫ��������ʽ�������Բⶨ�Ȼ�����Ʒ�Ĵ��ȡ���ش��������⣺
��1���ζ���Ӧ�ﵽ�յ�������ǣ� ��
��2��ʵʩ�ζ�����Һ�Գ� ��ѡ����ԡ��������ԡ����ԡ���Ϊ�ѣ����� ���ѧʽ���Լ��ɴ���һĿ�ġ�
��3�����յ㵽��֮ǰ�ĵζ������У����ֳ����������������Ag+���費�Ͼ���ҡ����ƿ�������ʹn (Cl��)�IJⶨ��� ��ѡ�ƫ�ߡ�����ƫ�͡�����Ӱ�족����
��4�������յ�����������ҡ����ƿ������ʱ���þ���ҡ�����Լ�����������������ʹn (Cl��)�ⶨֵƫ�ͣ���˵�����ߵ��ܽ�ȣ�AgCl AgSCN
���������������
��5������Fe3+����Һ��ָʾ������NH4SCN����Һ�ζ�ʣ���AgNO3ʱ����ȥ����Ũ�ȵ�NH4SCN��Һ20.0mL����ԭ�Ȼ�����Ʒ�Ĵ���Ϊ ��
A�������ʽṹ�����ʡ�ģ�飨12�֣�
��1��1s22s22p63s23p63d6��2�֣�
��2�����ӣ���λ������2�֣�
![]() ��3��1��9����1�֣�
��3��1��9����1�֣�
��4��C22- ����2�֣�
B����ʵ�黯ѧ��ģ�飨12�֣�
��1����Һ����ɫ��ΪѪ��ɫ��2�֣� ��2�����ԡ�HNO3����1�֣�
��3��ƫ�ߣ�2�֣� ��4���� ��2�֣�
��5��0.2mol��L�D1��2�֡�ȱ��λ��1�֣�
��6��79.3%��2�֣�
����:
A ��ï�����۵���173�棨��100��ʱ��ʼ���������е���249�棬������ˮ�������ڱ������ѵȷǼ����ܼ����ɼ���ï���۷е�;��з��Ӿ����ص㡣Fe2+�ṩ�չ����C5H5���ṩ�µ��Ӷԣ��Ӷ��γ���λ����

 �п������п��Ծ����ϵ�д�
�п������п��Ծ����ϵ�д� ��������״Ԫ��ϵ�д�
��������״Ԫ��ϵ�д�



 ���м������Ga���õ�������һ���������巢������������������ȫ���ҡ�
���м������Ga���õ�������һ���������巢������������������ȫ���ҡ�