��Ŀ����
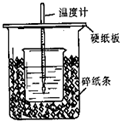
����ͼ��ʾװ�ý����к��Ȳⶨʵ�飬��ش����⣺
��1����С�ձ�֮����������ĭ���ϵ�������
��2��ʹ�ò�ȫ�������װ�ý���ʵ�飬ȡ50mL0.25mol/LH2SO4��Һ��50mL0.55mol/LNaOH��Һ��С�ձ��н����кͷ�Ӧ������ʵ���¶�ƽ������3.4�森��֪�кͺ����ɵ���Һ�ı�����Ϊ4.18J/��g?�棩����Һ���ܶȾ�Ϊ1g/cm3��ͨ������ɵ��к��ȡ�H=
H2SO4��aq��+NaOH��aq��=Na2SO4��aq��+H2O��l������H=-56.8KJ/mol
H2SO4��aq��+NaOH��aq��=Na2SO4��aq��+H2O��l������H=-56.8KJ/mol��
��3��ʵ��������60mL0.25mol?L-1H2SO4��Һ��50mL0.55mol?L-1NaOH��Һ���з�Ӧ��������ʵ����ȣ����ų�������
��1����С�ձ�֮����������ĭ���ϵ�������
���¡����ȡ�����ʵ������е�����ɢʧ
���¡����ȡ�����ʵ������е�����ɢʧ
����ʵ��װ���Ͽ���ͼ��ȱ�ٵ�һ�ֲ������������β���������
�����������
����2��ʹ�ò�ȫ�������װ�ý���ʵ�飬ȡ50mL0.25mol/LH2SO4��Һ��50mL0.55mol/LNaOH��Һ��С�ձ��н����кͷ�Ӧ������ʵ���¶�ƽ������3.4�森��֪�кͺ����ɵ���Һ�ı�����Ϊ4.18J/��g?�棩����Һ���ܶȾ�Ϊ1g/cm3��ͨ������ɵ��к��ȡ�H=
-56.8KJ/mol
-56.8KJ/mol
��H2SO4��NaOH��Ӧ���Ȼ�ѧ����ʽΪ| 1 |
| 2 |
| 1 |
| 2 |
��3��ʵ��������60mL0.25mol?L-1H2SO4��Һ��50mL0.55mol?L-1NaOH��Һ���з�Ӧ��������ʵ����ȣ����ų�������
�����
�����
�����ȡ���������ȡ����������к������
���
�����ȡ���������ȡ�����������50mL0.50mol?L-1�������H2SO4��Һ��������ʵ�飬��÷�Ӧǰ���¶ȵı仯ֵ��ƫС
ƫС
���ƫ����ƫС����������Ӱ�족������������1���к��Ȳⶨʵ��ɰܵĹؼ��DZ��¹������������ȼƵĹ������жϸ�װ�õ�ȱ��������
��2�����ݹ�ʽQ=��H=��Tcm�����㷴Ӧ���ʱ䣬�������Ȼ�ѧ����ʽ����д������д�Ȼ�ѧ����ʽ��
��3����Ӧ�ų����������������Լ�������Ķ����йأ��������к��ȵĸ����ʵ�����ش�
��2�����ݹ�ʽQ=��H=��Tcm�����㷴Ӧ���ʱ䣬�������Ȼ�ѧ����ʽ����д������д�Ȼ�ѧ����ʽ��
��3����Ӧ�ų����������������Լ�������Ķ����йأ��������к��ȵĸ����ʵ�����ش�
����⣺��1���к��Ȳⶨʵ��ɰܵĹؼ��DZ��¹�������С�ձ�֮����������ĭ���ϵ������ǣ����¡����ȡ�����ʵ������е�����ɢʧ���������ȼƵĹ����֪��װ�õ�ȱ�������ǻ��β������������ʴ�Ϊ�����¡����ȡ�����ʵ������е�����ɢʧ�����β�����������
��2��Q=��H=-��Tcm=-3.4��C��4.18J/��g?�棩��100g�T1421.2J=1.42KJ/mol����H=-Q/n��H2O��=-56.8KJ/mol���Ȼ�ѧ����ʽΪ��
H2SO4��aq��+NaOH��aq��=Na2SO4��aq��+H2O��l������H=-56.8KJ/mol���ʴ�Ϊ��
H2SO4��aq��+NaOH��aq��=Na2SO4��aq��+H2O��l������H=-56.8KJ/mol��
��3����Ӧ�ų����������������Լ�������Ķ����йأ�������60mL0.25mol?L-1H2SO4��Һ��50mL0.55mol?L-1NaOH��Һ���з�Ӧ��������ʵ����ȣ�����ˮ�������࣬���ų�������ƫ�ߣ������к��ȵľ���ǿ���ǿ�Ӧ����1molˮʱ�ų����ȣ��к�����ȣ��ִ��������ᣬ����������ȣ�������50mL0.50mol?L-1�������H2SO4��Һ��������ʵ�飬��÷�Ӧǰ���¶ȵı仯ֵ���С��
�ʴ�Ϊ������ȣ���ȣ�ƫС��
��2��Q=��H=-��Tcm=-3.4��C��4.18J/��g?�棩��100g�T1421.2J=1.42KJ/mol����H=-Q/n��H2O��=-56.8KJ/mol���Ȼ�ѧ����ʽΪ��
| 1 |
| 2 |
| 1 |
| 2 |
��3����Ӧ�ų����������������Լ�������Ķ����йأ�������60mL0.25mol?L-1H2SO4��Һ��50mL0.55mol?L-1NaOH��Һ���з�Ӧ��������ʵ����ȣ�����ˮ�������࣬���ų�������ƫ�ߣ������к��ȵľ���ǿ���ǿ�Ӧ����1molˮʱ�ų����ȣ��к�����ȣ��ִ��������ᣬ����������ȣ�������50mL0.50mol?L-1�������H2SO4��Һ��������ʵ�飬��÷�Ӧǰ���¶ȵı仯ֵ���С��
�ʴ�Ϊ������ȣ���ȣ�ƫС��
���������⿼��ѧ���й��к��ȵIJⶨ֪ʶ�����Ը�����ѧ֪ʶ���лش��ѶȲ���

��ϰ��ϵ�д�
 �ƸԴ��ž�ϵ�д�
�ƸԴ��ž�ϵ�д�
�����Ŀ
 ��2009?����ģ�⣩����ͼ��ʾװ�ý�������ʵ�飬�ܴﵽʵ��Ŀ���ǣ�������
��2009?����ģ�⣩����ͼ��ʾװ�ý�������ʵ�飬�ܴﵽʵ��Ŀ���ǣ������� ����ͼ��ʾװ�ý���ʵ�飬��A��μ���B�У�
����ͼ��ʾװ�ý���ʵ�飬��A��μ���B�У� ����ͼ��ʾװ�ý���ʵ�飬��A��μ���B�У�
����ͼ��ʾװ�ý���ʵ�飬��A��μ���B�У�