��Ŀ����
��13�֣������ڵ�����Ԫ��A��B��C��D��E��ԭ��������������A��B��C����Ԫ��ԭ�Ӻ�����Ӳ���֮����5��A��B��Ԫ��ԭ��������Ӳ��ϵ�����֮�͵���CԪ��ԭ��������Ӳ��ϵĵ�������BԪ��ԭ��������Ӳ��ϵĵ����������ĵ��Ӳ�����2����A��C�����γɻ�����CA3��B��D��ԭ������֮��Ϊ3�U4��Eԭ�������������ȴ�����������1����ش�
��1��E��Ԫ�����ڱ���λ��Ϊ���ߡ���
��2��A��B��C��Dԭ�Ӱ뾶�Ӵ�С��˳��Ϊ���ߡ��ߣ���Ԫ�ط��ţ�
��3����A��B����Ԫ����ɵķ����ж��֣�����һ�ַ��Ӻ�4��ԭ�ӣ��ҷ���Ϊֱ���͵ĶԳƽṹ��д�������ʽ�ߡ��ߡ�
��4������A��B��C��D����Ԫ����ɵ����Σ��ȿ��������ᷴӦ�ֿ�����NaOH�� Һ��Ӧ���仯ѧʽΪ�ߡ��ߡ�
Һ��Ӧ���仯ѧʽΪ�ߡ��ߡ�
��5����A��C��E����Ԫ����ɵĻ�����C2A6E2��һ����Ҫ�Ļ���ԭ�ϣ��������ӻ����������ˮ����Һ�����ԣ�ˮ��ԭ����NH4Cl���ơ�д���û������һ��ˮ�ⷴӦ�����ӷ���ʽ�ߡ��ߡ�
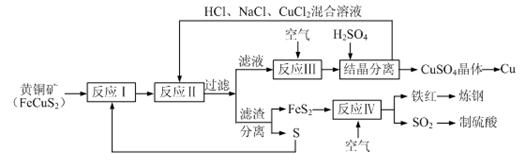
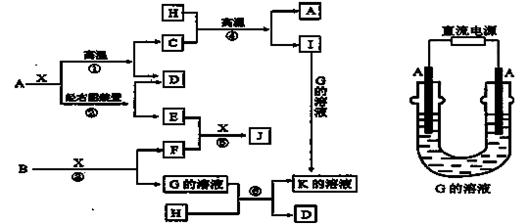
��6����̬��������ҽѧ������Ҫ����;��������Fe3O4�Ǵ������е���Ҫ���ӣ����Ʊ����̼�ʾ���£�
�ٽ�������CA3ͨ������ʵ�����ϵ�FeSO4��Fe2(SO4)3��Һ�У��������ּд���ù��̵������ӷ���ʽ�ߡ��ߡ�
��������Ӧ���ɵ����ּ�������ã��õ�Fe3O4���÷�Ӧ�Ļ�ѧ����ʽΪ�ߡ��ߡ�
��1��E��Ԫ�����ڱ���λ��Ϊ���ߡ���
��2��A��B��C��Dԭ�Ӱ뾶�Ӵ�С��˳��Ϊ���ߡ��ߣ���Ԫ�ط��ţ�
��3����A��B����Ԫ����ɵķ����ж��֣�����һ�ַ��Ӻ�4��ԭ�ӣ��ҷ���Ϊֱ���͵ĶԳƽṹ��д�������ʽ�ߡ��ߡ�
��4������A��B��C��D����Ԫ����ɵ����Σ��ȿ��������ᷴӦ�ֿ�����NaOH��
 Һ��Ӧ���仯ѧʽΪ�ߡ��ߡ�
Һ��Ӧ���仯ѧʽΪ�ߡ��ߡ���5����A��C��E����Ԫ����ɵĻ�����C2A6E2��һ����Ҫ�Ļ���ԭ�ϣ��������ӻ����������ˮ����Һ�����ԣ�ˮ��ԭ����NH4Cl���ơ�д���û������һ��ˮ�ⷴӦ�����ӷ���ʽ�ߡ��ߡ�
��6����̬��������ҽѧ������Ҫ����;��������Fe3O4�Ǵ������е���Ҫ���ӣ����Ʊ����̼�ʾ���£�
�ٽ�������CA3ͨ������ʵ�����ϵ�FeSO4��Fe2(SO4)3��Һ�У��������ּд���ù��̵������ӷ���ʽ�ߡ��ߡ�
��������Ӧ���ɵ����ּ�������ã��õ�Fe3O4���÷�Ӧ�Ļ�ѧ����ʽΪ�ߡ��ߡ�
��13�֣���1���������ڢ�A�� ��2��C��N��O��H��3��
��4��NH4HCO3 [��(NH4)2CO3]�� ��5��N2H62+ + H2O [N2H5��H2O]+ + H+
[N2H5��H2O]+ + H+
��6����Fe2+ + 2Fe3++8NH3 +8H2O = Fe(OH)2��+2Fe(OH)3��+8NH4+
��Fe(OH)2 +2Fe(OH)3 = Fe3O4 + 4H2O

��4��NH4HCO3 [��(NH4)2CO3]�� ��5��N2H62+ + H2O
 [N2H5��H2O]+ + H+
[N2H5��H2O]+ + H+��6����Fe2+ + 2Fe3++8NH3 +8H2O = Fe(OH)2��+2Fe(OH)3��+8NH4+
��Fe(OH)2 +2Fe(OH)3 = Fe3O4 + 4H2O
��

��ϰ��ϵ�д�
 ��ǰ����ϵ�д�
��ǰ����ϵ�д�
�����Ŀ
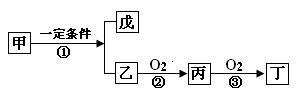
 ������Ϊȼ�ϵ�����ȼ�ϵ�ؽṹʾ��ͼ��ͼ��ʾ����ԭ��صĸ�����Ӧ����ʽΪ
������Ϊȼ�ϵ�����ȼ�ϵ�ؽṹʾ��ͼ��ͼ��ʾ����ԭ��صĸ�����Ӧ����ʽΪ 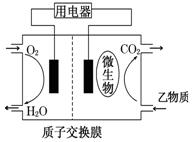

 3++
3++  Fe3��+ H2O��δ��ƽ��
Fe3��+ H2O��δ��ƽ�� ��D��F��N��OΪ��ɫ���壬E���³�ѹ��Ϊ��ɫ��ζ��Һ�壬N��H��LΪ���г����ĵ��ʣ�IΪ��������ǿ�ᣬM����ɫ��ӦΪ��ɫ����Ӧ�ٳ���������F�ļ��顣
��D��F��N��OΪ��ɫ���壬E���³�ѹ��Ϊ��ɫ��ζ��Һ�壬N��H��LΪ���г����ĵ��ʣ�IΪ��������ǿ�ᣬM����ɫ��ӦΪ��ɫ����Ӧ�ٳ���������F�ļ��顣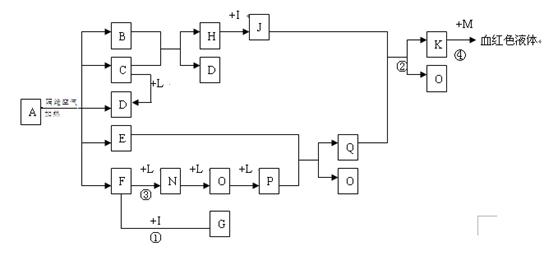

 _______��
_______��