��Ŀ����
6�����������ķ�չ���̣�Ҳ�ǻ�ѧ���ʵ���ʶ�ͷ��ֵ����̣�������������ء���ù�ء������Ҵ���������������ϩ�����������17�֡����ӡ��ı����������磮��1����ԭ���ڻ�̬ʱ���۵��ӣ���Χ���ӣ��Ų�ʽΪ3d64s2��
��2���������NO3-�Ŀռ乹��Ϊƽ���������Σ�д����NO3-��Ϊ�ȵ������һ�ַǼ��Է��ӻ�ѧʽBF3[SO3�� g����BBr3��]��
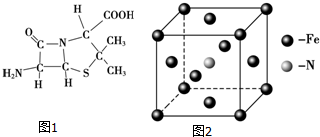
��3��6������ù����Ľṹ��ͼ1��ʾ�����в���sp3�ӻ���ԭ����C��N��O��S��
��4������˵����ȷ����a������ĸ��ţ���
a���Ҵ����Ӽ���γ������������е���������
b���ٵ����������[W��CO��5OH]-�ܴ��̶�CO2����������������-1��
c������ϩ��
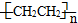 ����������5n���Ҽ�
����������5n���Ҽ�d�����±������ݿ�ȷ���ڷ�ӦSi��s��+O2��g���TSiO2��s���У�ÿ����60g SiO2�ų�������Ϊ��2c-a-b�� kJ
| ��ѧ�� | Si-Si | O�TO | Si-O |
| ���ܣ�kJ•mol-1�� | a | b | c |
���� ��1������26��Ԫ�أ���̬ԭ�Ӻ�����26�����ӣ�3d��4s�ܼ��ϵ�������۵��ӣ����ݹ���ԭ����д���̬�۵����Ų�ʽ��
��2�����ݼ۲���ӶԻ�������ȷ�����ӿռ乹�ͣ�ԭ�Ӹ�����ȼ۵�������ȵ����ǵȵ����壬������������غϵķ����ǷǼ��Է��ӣ�
��3�����ݼ۲���ӶԻ�������ȷ��̼ԭ���ӻ���ʽ��
��4��a����Ĵ���Ӱ�����ʵ��۷е㣻
b���ݻ������л��ϼ۵Ĵ�����Ϊ0���㣻
c����ϩ��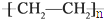 ���������У�6n-1�����Ҽ���
���������У�6n-1�����Ҽ���
d.60gSiO2�����ʵ���=$\frac{60}{60}$��һ����ԭ�Ӻ���4����������
��5�����Ͱ�����640��ɷ����û���Ӧ���������͵����������þ�̯��ȷ���������Ļ�ѧʽ�������¶ȡ���Ӧ���������д����Ӧ����ʽ��
������������ⳤΪxcm���������������Խ���Ϊ$\sqrt{2}$xcm�������������Feԭ�Ӽ�ľ���Ϊscm������$\frac{\sqrt{2}}{2}$x=s����x=$\sqrt{2}$scm�����ݦ�=$\frac{m}{V}$���㾧���ܶȣ�
��� �⣺��1������26��Ԫ�أ���̬ԭ�Ӻ�����26�����ӣ�3d��4s�ܼ��ϵ�������۵��ӣ����ݹ���ԭ��֪��۵����Ų�ʽΪ��3d64s2���ʴ�Ϊ��3d64s2��
��2��NO3-��Nԭ�Ӽ۲���Ӷ�=3+$\frac{1}{2}$��5+1-3��2��=3���Ҳ����µ��Ӷԣ�������ƽ�������νṹ��
��N03-��Ϊ�ȵ������һ�ַǼ��Է����У�BF3[SO3�� g����BBr3��]��
�ʴ�Ϊ��ƽ���������Σ�BF3[SO3�� g����BBr3��]��
��3��ֻҪ���۵����µ��ӶԵĺ���4��ԭ�ӾͲ�ȡsp3�ӻ�������ͼƬ֪������sp3�ӻ���ԭ����C��N��O��S���ʴ�Ϊ��C��N��O��S��
��4��a���Ҵ����Ӽ���γ�������������в������������Ĵ��ڵ����Ҵ��ķе����ߣ������Ҵ��ķе��������ߣ�����ȷ��
b�����ݻ������л��ϼ۵Ĵ�����Ϊ0֪���ٵ����������[W��CO��5OH]-������0�ۣ��ʴ���
c������ϩ��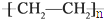 ���������У�6n-1�����Ҽ����ʴ���
���������У�6n-1�����Ҽ����ʴ���
d���ɱ�������֪��ӦSi��s��+O2��g��=SiO2��s���У�ÿ����60gSiO2�ų�������Ϊ��4c-2a-b��kJ���ʴ���
��ѡa��
��5���þ�������ԭ�Ӹ���=8��$\frac{1}{8}$+6��$\frac{1}{2}$=4����ԭ�Ӹ�����1�����Ե������Ļ�ѧʽ��Fe4N�����Ͱ�����640��ɷ����û���Ӧ���������͵����������Ը÷�Ӧ����ʽΪ��8Fe+2NH3$\frac{\underline{\;640��\;}}{\;}$2Fe4N+3H2��
������������ⳤΪxcm���������������Խ���Ϊ$\sqrt{2}$xcm�������������Feԭ�Ӽ�ľ���Ϊscm������$\frac{\sqrt{2}}{2}$x=s����x=$\sqrt{2}$scm�����ݦ�=$\frac{m}{V}$=$\frac{\frac{56��4+14}{N{\;}_{A}}}{��\sqrt{2}s��{\;}^{3}}$=$\frac{{119\sqrt{2}}}{{2{s^3}{N_A}}}$��
�ʴ�Ϊ��8Fe+2NH3$\frac{\underline{\;640��\;}}{\;}$2Fe4N+3H2��$\frac{{119\sqrt{2}}}{{2{s^3}{N_A}}}$��
���� ���⿼����ۺϣ��������Ų�ʽ����д���ӻ���ʽ���жϡ������ļ��㶼��ѧϰ�ص㣬�ѵ��ǣ�4��cdѡ�cѡ����������ѧ���ɷ�������dѡ������ȷ�衢��������Ŀռ�ṹ�ǽ��ѡ���ǹؼ����Ѷ��еȣ�

| A�� | �ۢ� | B�� | �٢� | C�� | �٢� | D�� | �٢ۢ� |
| A�� | ��̼������Һ����������������2CO32-+SO2+H2O�T2HCO3-+SO32- | |
| B�� | ��̼��������Һ�м����������ƣ�HCO3-+OH-�TCO2��+H2O | |
| C�� | ��FeCl3��Һ�м���ͭƬ��Fe3++Cu�TFe2++Cu2+ | |
| D�� | ��NaHSO4��Һ�еμӹ���Ba��OH��2��Һ��2H++SO42-+Ba2++2OH-�TBaSO4��+2H2O |
| A�� | ����ʯ�����ᷴӦ��CaCO3+2H+=Ca2++H2O+CO2�� | |
| B�� | ���������Ʒ�����ˮ�У�Na+2H2O=Na++2OH-+H2�� | |
| C�� | �����������������Һ��Ӧ��SiO2+2OH-=SiO${\;}_{3}^{2-}$+H2O | |
| D�� | ����ʯ��ˮͨ�����CO2���ܷ�Ӧ��CO2+OH-=HCO${\;}_{3}^{-}$ |
| A�� | ���³�ѹ�£���������̬���嵥�ʳ�Һ̬ | |
| B�� | �����ǻӷ����ᣬ�������ѻӷ��Ե��� | |
| C�� | ϡ������һ���ѷ�����ѧ��Ӧ | |
| D�� | �����ڳ����º��ȶ�����ѧ���ʲ����� |
| ѡ�� | ʵ������ | ���� |
| A | ij���ʵ�ˮ��Һ��ʹ��ɫʯ����ֽ���� | ������һ���ǰ��� |
| B | ij���ʵ�ˮ��Һ�м������������ɫ��ζ���� | ����Һһ�����д���̼������� |
| C | �ⶨͬŨ��Na2CO3 ��Na2SO4 ��Һ��pH��ǰ�ߴ��ں��� | ̼Ԫ�طǽ����������� |
| D | ���ˮ����μ���1��2mL FeCl3������Һ�����貢������г��ֻ��� | �Ʊ�Fe��OH��3���� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | SO2�����Ա�I2ǿ | |
| B�� | ��ԭ�ԣ�I-��SO2��Cl- | |
| C�� | Cl2������I-��SO2 | |
| D�� | �����Һ����Һһ������Cl-��SO42-��I2 |