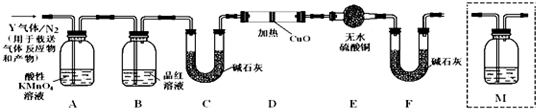
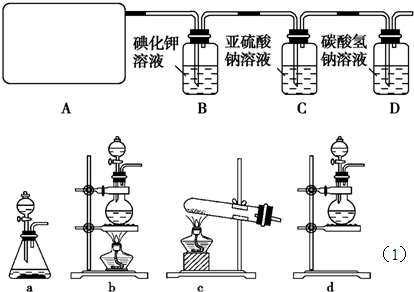
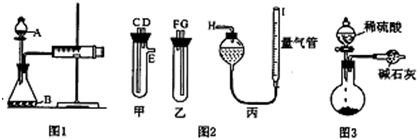
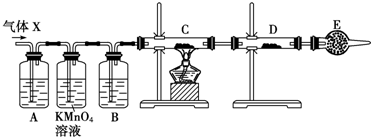
��Ŀ����
ijͬѧ��ȡ��4.0gNaOH���壬������100mL��Һ���ش��������⣺
��1����ʵ���У��õ�����ȷ����������ֲ���������
��2�����������ж���õ�������������ʵ���е�������
��3�����淶��ʵ������ᵼ��ʵ���������������в�����ʵ������Ӱ�죨�ƫ����ƫС�����䡱����
�ٶ���ʱ���������ϵĿ̶���
�ڶ��ݺ�����ƿ��ҡ�Ⱥ��÷���Һ����ڿ̶��ߣ������ּ�������ˮ���̶���
��1����ʵ���У��õ�����ȷ����������ֲ���������
��Ͳ
��Ͳ
��100mL����ƿ
100mL����ƿ
����2�����������ж���õ�������������ʵ���е�������
���衢������
���衢������
����3�����淶��ʵ������ᵼ��ʵ���������������в�����ʵ������Ӱ�죨�ƫ����ƫС�����䡱����
�ٶ���ʱ���������ϵĿ̶���
ƫС
ƫС
���ڶ��ݺ�����ƿ��ҡ�Ⱥ��÷���Һ����ڿ̶��ߣ������ּ�������ˮ���̶���
ƫС
ƫС
����������1������ʵ������IJ����Լ�ÿ��������Ҫ����ȷ����Ӧ����������Ȼ��ȷ���õ�����ȷ����������ֲ���������
��2�����ݹ����ܽ����ò���������ӿ��ܽ⣬��ת����Һʱ�ò�����������
��3������C=
�жϣ����nƫС��Vƫ����������Һ��Ũ��ƫ�ͣ����nƫ���VƫС����������ҺŨ��ƫ��
��2�����ݹ����ܽ����ò���������ӿ��ܽ⣬��ת����Һʱ�ò�����������
��3������C=
| n |
| V |
����⣺��1�����������м��㡢������ϡ�͡���Һ��ϴ����Һ�����ݡ�ҡ�ȵȲ�������������ƽ��ҩ�׳�ȡ�������ƣ����ձ���ϡ�ͣ�������Ͳ��ˮ�����ò��������裬��ȴ�����º�ת�Ƶ�100mL����ƿ�У����ò�����������ϴ��2-3�Σ���ϴ��Һת�Ƶ�����ƿ�У���ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμ�����Һ��Һ����̶���ˮƽ���У��Ǻ�ƿ���ߵ�ҡ�ȣ�������Ҫ������Ϊ��������ƽ��ҩ�ס���Ͳ���ձ�����������100mL����ƿ����ͷ�ιܣ��õ�����ȷ����������ֲ�����������Ͳ��100mL����ƿ��
�ʴ�Ϊ����Ͳ��100mL����ƿ��
��2��NaOH�����ܽ����ò���������ӿ��ܽ⣬��ת����Һʱ�ò��������������Բ�����ʵ���е������н��衢������
�ʴ�Ϊ�����衢������
��3���ٶ���ʱ���������ϵĿ̶��ߣ���Һ�����ƫ����������ҺŨ��ƫС��
�ڶ��ݺ�����ƿ��ҡ�Ⱥ��÷���Һ����ڿ̶��ߣ������ּ�������ˮ���̶��ߣ�Һ��������������Һ�����ƫ����������ҺŨ��ƫС��
�ʴ�Ϊ����ƫС ��ƫС��
�ʴ�Ϊ����Ͳ��100mL����ƿ��
��2��NaOH�����ܽ����ò���������ӿ��ܽ⣬��ת����Һʱ�ò��������������Բ�����ʵ���е������н��衢������
�ʴ�Ϊ�����衢������
��3���ٶ���ʱ���������ϵĿ̶��ߣ���Һ�����ƫ����������ҺŨ��ƫС��
�ڶ��ݺ�����ƿ��ҡ�Ⱥ��÷���Һ����ڿ̶��ߣ������ּ�������ˮ���̶��ߣ�Һ��������������Һ�����ƫ����������ҺŨ��ƫС��
�ʴ�Ϊ����ƫС ��ƫС��
���������⿼��һ�����ʵ���Ũ����Һ�����ƣ��ѶȲ���ע��ʵ�鲽�衢���������á�������Ϊ�״��㣮

��ϰ��ϵ�д�
 ͬ��ѧ��һ�ζ���ϵ�д�
ͬ��ѧ��һ�ζ���ϵ�д� �����ܾ�ϵ�д�
�����ܾ�ϵ�д� ���ƿ�����ϵ�д�
���ƿ�����ϵ�д� ���¿쳵����������ϵ�д�
���¿쳵����������ϵ�д�
�����Ŀ