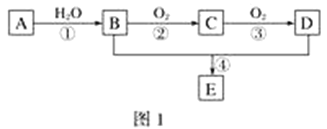
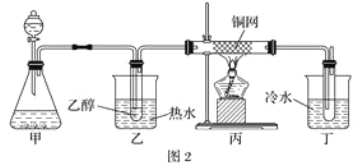
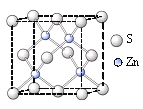
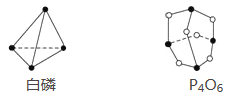��Ŀ����
����Ŀ��������ͬѧ����Ϥ�����ʣ�
��O2����H2O2����MgCl2����H2SO4����Na2CO3��NH4Cl����CO2����Ne����Na2O2����NaOH
(1)��Щ�����У�ֻ���й��ۼ�����__________________________________(����ţ���ͬ)��ֻ�������Ӽ�����___________________________���Ⱥ��м��Թ��ۼ��ֺ������Ӽ�����_____________________________���Ⱥ��зǼ��Թ��ۼ��ֺ������Ӽ�����________________________�����ڻ�ѧ������_________________________________��
���ڹ��ۻ��������_________________________________��
(2)д���������ʵĽṹʽ
��O2 _______________��H2O2_______________��CO2_______________
(3)д���������ʵĵ���ʽ
��NH4Cl ______________��Na2O2 _______________����NaOH_______________
(4)�õ���ʽ��ʾ�������ʵ��γ�
��O2_______________________________________________________________
��MgCl2____________________________________________________________
���𰸡��٢ڢܢ� �� �ݢޢ� �� �� �ڢܢ� O=O H-O-O-H O=C=O 

![]()
![]() +
+![]()
![]()
![]()
![]()
��������
(1)��O2��������ԭ�ӹ��ɣ�ֻ���Ǽ��Թ��ۼ�������H2O2�ɷǽ���Ԫ����ɣ��ṹ��ʽ��H-O-O-H���Ⱥ��м��Թ��ۼ��ֺ��зǼ��Թ��ۼ�����MgCl2�ɻ��ý����ͻ��÷ǽ���Ԫ����ɣ�ֻ�������Ӽ�������H2SO4�ɷǽ���Ԫ����ɣ�ֻ�����Թ��ۼ�������Na2CO3�������Ӻ�̼������ӹ��ɣ��������Ӽ��ͼ��Թ��ۼ�����NH4Cl��笠����Ӻ������ӹ��ɣ��������Ӽ��ͼ��Թ��ۼ�������CO2�ɷǽ���Ԫ����ɣ�ֻ�����Թ��ۼ�������Ne�ǵ�ԭ�ӷ��ӣ�������ѧ��������Na2O2�������Ӻ��������ӹ��ɣ��������Ӽ��ͷǼ��Թ��ۼ�������NaOH�������Ӻ����������ӹ��ɣ��������Ӽ��ͼ��Թ��ۼ�����Щ�����У�ֻ���й��ۼ�����O2��H2O2��H2SO4��CO2�����٢ڢܢߣ�ֻ�������Ӽ�����MgCl2����ѡ�ۣ��Ⱥ��м��Թ��ۼ��ֺ������Ӽ�����Na2CO3��NH4Cl��NaOH����ѡ�ݢޢ⣻�Ⱥ��зǼ��Թ��ۼ��ֺ������Ӽ�����Na2O2��ѡ������ڻ�ѧ������Ne ��ѡ�ࣻ���ڹ��ۻ��������H2O2��H2SO4��CO2��ѡ�ڢܢߡ�
(2)O2 ��֮�ں���˫����O2�Ľṹʽ��O=O��H2O2�Ľṹʽ��H-O-O-H��CO2�Ľṹʽ��O=C=O��
(3)NH4Cl �����ӻ��������ʽ��![]() ��Na2O2 �����ӻ��������ʽ��
��Na2O2 �����ӻ��������ʽ��![]() ����NaOH�����ӻ��������ʽ��
����NaOH�����ӻ��������ʽ��![]() ��
��
(4)������ԭ��ͨ��˫���γ�O2���γɹ��̿ɱ�ʾΪ![]() +
+![]()
![]()
![]() ��
��
MgCl2�����ӻ����MgCl2���γɹ��̿ɱ�ʾΪ![]() ��
��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�����Ŀ�����з�̪��Ba(OH)2��Һ�Ժ�ɫ����������Һ�зֱ�μ�X��Һ����������������˵������ȷ����
��� | װ�� | X��Һ | ���� |
I |
| ���� | ��Һ��ɫ��ȥ����������������û�����Ա仯 |
II | Na2SO4 | ��Һ��ɫ���䣬�а�ɫ�������ɣ���������û�����Ա仯 | |
III | H2SO4 | ��Һ��ɫ��ȥ���а�ɫ�������ɣ������䰵 |
A. ʵ��I����Һ��ɫ��ȥ��˵�������˷�ӦH+ + OH- =H2O
B. ʵ��II����Һ��ɫ���䣬�ҵ�������û�����Ա仯��˵����Һ����Ȼ�����д�����Na+��OH-
C. ʵ��III����Һ��ɫ��ȥ���а�ɫ�������ɣ������䰵��˵�������˷�ӦBa2+ + 2OH- + 2H+ + SO42- == BaSO4��+ 2H2O
D. ��ʵ��II��Na2SO4��Һ����CuSO4��Һ��������ԭʵ��II�е�������ͬ