��Ŀ����
��ҵ�ϣ���������Ϊԭ���Ʊ��������ѵ�ij������������ͼ��ʾ����������Ҫ�ɷ�
Ϊ��������( FeTiO3)������һ������Ԫ���ڷ绯�����л�ת��Ϊ+3�ۡ�

���������գ�
��1��������У������۽�Fe3��ת��ΪFe2���ķ�Ӧ�����ӷ���ʽΪ____��
��2��������У�ʵ�ֻ����ķ������������ʵ�____������ĸ��ţ���
a���۷е���� b�ܽ��Բ��� c�����ԡ���ԭ�Բ���
��3������ڡ��ۡ����У�������еIJ�����____����������ƣ���
��4�����������������еķ�Һ�����̿���Ҫ�ɷ�MnO2����Ӧ���������̣���Ӧ�����ӷ���ʽΪ
��5��������ͼװ�ã�ʯī������������������������CaF2��CaO������ʣ��ɻ�ý����ƣ�������Ϊ��ԭ�����ɻ�ԭ���������Ƹ������ѡ�
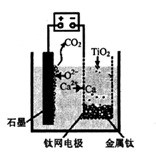
�������������ķ�ӦΪ________��
�����Ʊ�������ǰ��CaO���������䣬��ԭ����____��
��6��_Tҵ����4.0���������Ƶ�1. 6�ֵĶ������ѣ�������������Ԫ�ص�����������____��
������������������û����ʧ��
Ϊ��������( FeTiO3)������һ������Ԫ���ڷ绯�����л�ת��Ϊ+3�ۡ�

���������գ�
��1��������У������۽�Fe3��ת��ΪFe2���ķ�Ӧ�����ӷ���ʽΪ____��
��2��������У�ʵ�ֻ����ķ������������ʵ�____������ĸ��ţ���
a���۷е���� b�ܽ��Բ��� c�����ԡ���ԭ�Բ���
��3������ڡ��ۡ����У�������еIJ�����____����������ƣ���
��4�����������������еķ�Һ�����̿���Ҫ�ɷ�MnO2����Ӧ���������̣���Ӧ�����ӷ���ʽΪ
��5��������ͼװ�ã�ʯī������������������������CaF2��CaO������ʣ��ɻ�ý����ƣ�������Ϊ��ԭ�����ɻ�ԭ���������Ƹ������ѡ�

�������������ķ�ӦΪ________��
�����Ʊ�������ǰ��CaO���������䣬��ԭ����____��
��6��_Tҵ����4.0���������Ƶ�1. 6�ֵĶ������ѣ�������������Ԫ�ص�����������____��
������������������û����ʧ��
��1�� 2Fe3++Fe=3Fe2+ (2��) ��2�� b (2��) ��3�� ���� (2��)
��4�� MnO2+2Fe2++4H+=Mn2++2Fe3++2H2O (2��)
��5���� 2O2-��4e-+C=CO2�� (2��)
�� �Ʊ�TiO2ʱ�����۷�����Ӧ��2CaO
 2Ca+O2����2Ca+ TiO2="Ti" +2CaO����֪CaO�����������(3��) �������𰸸��֣�
2Ca+O2����2Ca+ TiO2="Ti" +2CaO����֪CaO�����������(3��) �������𰸸��֣���6�� 24% (2��)
�����������1�����𰸣�2����FeSO4��7H2O����Һ�з�������������������ܽ��ԵIJ��죨3������ڡ��ۡ��ܾ��ǹ�Һ���루4����Һ�к���H+��Fe2+���л�ԭ�ԣ���MnO2����������ԭ��Ӧ��5����ͼ��֪��O2-�������ŵ磬ʯī�缫���뷴Ӧ������CO2
��6��Ti ~ TiO2
48 80
m= 0.96t 1.6t
������������Ԫ�ص�����������0.96��4.0��100%=24%

��ϰ��ϵ�д�
�����Ŀ

 2I��+S4O62����
2I��+S4O62����

 2H2(g)+S2(g),��ƽ�ⳣ������ʽΪK=��
2H2(g)+S2(g),��ƽ�ⳣ������ʽΪK=��