��Ŀ����
����Ŀ����1��ͬ��ͬѹ�µ�������ͬ�����зֱ�װ��O2��O3���壬����ƿ�����з�����֮����______��ԭ����֮����______������֮����______���ܶ�֮����______
��2�������������ļ��������У����з�����������_________������ԭ����������_________����״�������������____________��
�� 1gH2�� �� 2.408��1023��CH4 ���� 10.8gH2O�� �� ��״����6.72LCO2
��3����NA��ʾ�����ӵ����������agij���庬�еķ�������b����cg�������ڱ�״���µ������_________________���ú�NA��ʽ�ӱ�ʾ����
��4������100 mL 1mol/L�ĵ�ϡH2SO4��Һ����Ҫ����Ͳ��ȡ��������Ϊ98%��ŨH2SO4���ܶ�Ϊ1.84g/cm3�������Ϊ______________��
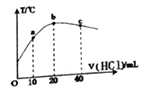
���𰸡�1��12��32��32��3�ۢڢ�![]() 5.4
5.4
��������
ͬ��ͬѹ�����������Ħ�������ȣ�����n=![]() =
=![]() =
=![]() ��Ϸ��ӹ��ɼ�����
��Ϸ��ӹ��ɼ�����
��1��ͬ��ͬѹ�����������Ħ�������ȣ���n=![]() =
=![]() =
=![]() ��֪������ķ�������ȣ���ֵΪ1��1����ԭ����Ϊ2��3�������ȵ���Ħ������֮�ȣ�Ϊ2��3������
��֪������ķ�������ȣ���ֵΪ1��1����ԭ����Ϊ2��3�������ȵ���Ħ������֮�ȣ�Ϊ2��3������![]() ��֪���ܶ������������ȣ����ܶ�֮��Ϊ2��3��
��֪���ܶ������������ȣ����ܶ�֮��Ϊ2��3��
��2����n��H2��=![]() =0.5mol��V��H2��=0.5mol��22.4L/mol=11.2L��ԭ�ӵ����ʵ���Ϊ1mol��
=0.5mol��V��H2��=0.5mol��22.4L/mol=11.2L��ԭ�ӵ����ʵ���Ϊ1mol��
��n��CH4��=![]() =0.4mol��V��CH4��=0.4mol��22.4L/mol=8.96L��ԭ�ӵ����ʵ���Ϊ2mol��
=0.4mol��V��CH4��=0.4mol��22.4L/mol=8.96L��ԭ�ӵ����ʵ���Ϊ2mol��
��n��H2O��=![]() =0.6mol��ԭ�ӵ����ʵ���Ϊ1.8mol�����ԼΪ10.8mL��
=0.6mol��ԭ�ӵ����ʵ���Ϊ1.8mol�����ԼΪ10.8mL��
��n��CO2��=![]() =0.3mol��ԭ�ӵ����ʵ���Ϊ0.9mol��
=0.3mol��ԭ�ӵ����ʵ���Ϊ0.9mol��
���ʵ���Խ������Խ�࣬�����Ǣۣ�ԭ�ӵ����ʵ���Խ�࣬ԭ�Ӹ���Խ�࣬����Ϊ�ڣ���״������������Ǣ���
��3��a��ij�����к��еķ�����Ϊb����c�����庬�еķ�����Ϊ![]() ��c�˸���������ʵ���Ϊ
��c�˸���������ʵ���Ϊ![]() =
=![]() mol���ڱ�״����Vm=22.4L/mol����cg����������Ϊ
mol���ڱ�״����Vm=22.4L/mol����cg����������Ϊ![]() mol��22.4L/mol=
mol��22.4L/mol=![]() L��
L��
��4���ܶ�Ϊ1.84gmL-1����������Ϊ98%��Ũ��������ʵ���Ũ��Ϊ![]() =18mol/L������ҪŨ�������ΪV����������Һϡ��ǰ���������ʵ����ʵ�������ã�V��18.4mol/L=1molL-1��100mL����ã�V=5.4mL��
=18mol/L������ҪŨ�������ΪV����������Һϡ��ǰ���������ʵ����ʵ�������ã�V��18.4mol/L=1molL-1��100mL����ã�V=5.4mL��