��Ŀ����
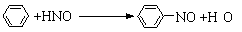
���Ȼ���(TiCl4)����ȡ���칤ҵ���ϡ����ѺϽ����Ҫԭ�ϡ�ʵ������TiO2��CCl4Ϊԭ����ȡҺ̬TiCl4��װ����ͼ��ʾ�����ּг�װ��ʡ�ԣ���
��֪���й����ʵ��������±���
���� | �۵�/�� | �е�/�� | ���� |
CCl4 | -23 | 76 | ��TiCl4���� |
TiCl4 | -25 | 136 | ����ʪ����������������550 ��ʱ�ܱ��������� |
��ش��������⣺
��1������A��������________������A��ʢװ���Լ���___________��
��2��CCl4�ĵ���ʽΪ__________��
��3��װ��C��Ӳ�ʲ����з�Ӧ�Ļ�ѧ����ʽΪ__________________��
��4��TiCl4����ʪ���������TiO2����Ӧ�Ļ�ѧ����ʽΪ________________��
��5������N���ռ�����������Ҫ��___________��д��ѧʽ�����Ӳ����з����TiCl4��ʵ�鷽����_____________��
��6��TiCl4������TiO2����̿�������ڼ����������Ƶã����������Ϊ2��1��CO��CO2������壬�÷�Ӧ���������뻹ԭ�������ʵ���֮��Ϊ________�������ʵ�鷽����֤������������CO:_______________��
 ��ѧʵ����ϵ�д�
��ѧʵ����ϵ�д�һ���¶��£���1L�����ܱ������м���lmol��N2(g)��3molH2(g)������Ӧ��N2(g)+3H2(g)  2NH3(g) ��H<0�� NH3�����ʵ�����ʱ��Ĺ�ϵ���±���ʾ������˵���������
2NH3(g) ��H<0�� NH3�����ʵ�����ʱ��Ĺ�ϵ���±���ʾ������˵���������
ʱ��(min) | 0 | t1 | t2 | t3 |
NH3���ʵ���(mol) | 0 | 0.2 | 0.3 | 0.3 |
A. 0〜t1min��v(NH3)=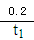 mol��L-1��min-1
mol��L-1��min-1
B. �����¶ȣ���ʹ����Ӧ���ʼ�С���淴Ӧ��������ƽ������
C. N2(g)+3H2(g)  2NH3(g)�Ļ��С��2NH3(g)
2NH3(g)�Ļ��С��2NH3(g)  N2(g)+3H2(g)�Ļ��
N2(g)+3H2(g)�Ļ��
D. t3ʱ�ټ���1mol��N2(g)��3molH2(g)����Ӧ����ƽ��ʱ��c(N2)>0.85mol��L-1


 H3O++OH����H3O+ͨ����дΪH+����
H3O++OH����H3O+ͨ����дΪH+����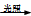 CH3Cl + HCl
CH3Cl + HCl CH2BrCH2Br
CH2BrCH2Br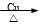 2CH3CHO + 2H2O
2CH3CHO + 2H2O