��Ŀ����
ij��ѧ��ȤС��ѡ������װ�ú�ҩƷ��ȡ������Ȳ�������й���Ȳ���ʵ�̽�����Իش��������⡣
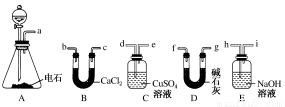
��1��A����ȡ��Ȳ�Ļ�ѧ����ʽΪ________________��
��2������Ȳʱ��������Һ©���Ļ�����ʹˮ�������µ�ԭ����_________________��
��3���õ�ʯ�Ƶõ���Ȳ�г�����H2S��PH3�����ʣ���ȥ����Ӧѡ��____________(����ţ���ͬ)װ�ã�������Ȳ���ѡ��________װ�á�
��4��Ϊ��̽����Ȳ��HBr�����ӳɷ�Ӧ����йز����������ʵ�飺

�ٲ���b��������________��
���л���������ܺ��е�������________(д�ṹ��ʽ)��
 Ӧ�����������Ĵ���ѧ������ϵ�д�
Ӧ�����������Ĵ���ѧ������ϵ�д����г��ӷ���������ǣ� ��
ѡ�� | ���ᴿ������ | ���� | �����Լ� | ���ӷ��� |
A | CH4��g�� | C2H4 ��g�� | ��ˮ��ŨH2SO4 | ϴ�� |
B | NH4Cl��aq�� | Fe3����aq�� | ��ˮ | ����pH��7��8���ٹ��� |
C | C | CuO��s�� | ϡ���� | ���� |
D | Na2CO3��s�� | NaHCO3 ��s�� | ���� | ���� |
ijͬѧ��������ʵ�飺
װ�� | ���� | ���� |
| ��ʢ��Ũ������ձ�A����ʢ�е���KI��Һ���ձ�C�У�Ȼ��ͭƬ�����ձ�A���������ձ�B��ס | �ձ�AҺ���Ϸ��������ִ�������ɫ���壻һ��ʱ�����ɫ������ʧ���ձ�A��C�е�Һ�嶼�����ɫ |
����˵����������
A���ձ�A�з�����Ӧ��3Cu+8HNO3===3Cu��NO3��2+2NO��+4H2O
B������ɫ������ʧֻ��NO2���ձ�C�е�KI������Ӧ�й�
C���ձ�C����Һ����ֻ��NO2��C����Һ������Ӧ�й�
D������ͭƬ������Ƭ����C�е�Һ��Ҳ���ܱ���


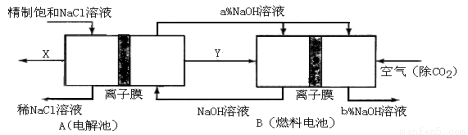
 ͼ��X��Y�ֱ���_____________��_____________���ѧʽ���������Ƚ�ͼʾ������������������a����b���Ĵ�С ��
ͼ��X��Y�ֱ���_____________��_____________���ѧʽ���������Ƚ�ͼʾ������������������a����b���Ĵ�С �� ������˵����ȷ����
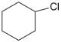
������˵����ȷ����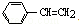 ���к��е�C=C��Ϊ4NA
���к��е�C=C��Ϊ4NA
 B��
B��  C��
C��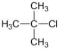 D��
D��
 u��s��
u��s��