��Ŀ����
�������������ĸ���Ӧ������������á���S+Hg�T�THgS����3S+2Al![]() Al2S3��
Al2S3��
��S+2KNO3+3C![]() K2S+N2��+3CO2����
K2S+N2��+3CO2����
��3S+6KOH�T�T2K2S+K2SO3+3H2O�����Тٳ������������ڹ���������Ⱦ���������Ʊ�Al2S3�������ҹ��Ŵ��Ĵ���֮һ�����ڻ�ҩ��һ�������ľ̿���ı�ը��Ӧ��������ϴ�������Թܵ������ڱڵIJ�����ķ���֮һ��
�����ڷ�Ӧ�еļ�̬�仯���飬����________������������ͬ��������Ҫ�ķ���_______
_______________________________________________________________________________
�𰸣�
������
������
| �� ��Ӧ�٢ڢ���������������������������������ǻ�ԭ������������������ԭ��Ӧ��
|

��ϰ��ϵ�д�
�����Ŀ
��֪��H2��g��+
O2��g���TH2O��l����H1=-285.8kJ?mol-1��
H2��g���TH2��l����H2=-0.92kJ?mol-1��
O2��g���TO2��l����H3=-6.84kJ?mol-1��
H2O��g���TH2O��l����H4=-44kJ?mol-1��
����˵����ȷ���ǣ�������
| 1 |
| 2 |
H2��g���TH2��l����H2=-0.92kJ?mol-1��
O2��g���TO2��l����H3=-6.84kJ?mol-1��
H2O��g���TH2O��l����H4=-44kJ?mol-1��
����˵����ȷ���ǣ�������
| A�������ĸ���Ӧ�������ȷ�Ӧ | ||
| B��1molҺ̬H2����������1mol��̬H2������ | ||
| C��H2��ȼ���ȣ���H��Ϊ-285.8kJ?mol-1 | ||
D�������Һ��ȼ�յ��Ȼ�ѧ����ʽΪH2��l��+
|
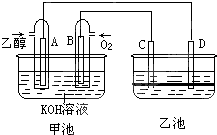 ��ͼ�׳غ��ҳ��е��ĸ��缫���Ƕ��Բ��ϣ��ҳ���Һ�ֲ㣬�ϲ���ҺΪ����Һ�������ԣ������ͼʾ�ش��������⣺
��ͼ�׳غ��ҳ��е��ĸ��缫���Ƕ��Բ��ϣ��ҳ���Һ�ֲ㣬�ϲ���ҺΪ����Һ�������ԣ������ͼʾ�ش��������⣺