��Ŀ����
����Ŀ��ʯ�Ͳ�Ʒ�к���H2S��COS��CH3SH�ȶ����л�����ʯ�ͻ�����������������������ش�����������
��1��COS�ĵ���ʽ��_______________��
��2����֪�Ȼ�ѧ����ʽ����2H2S(g)+SO2(g)=3S(s)+2H2O(l) ��H=-362kJ��mol-1
��2H2S(g)+3O2(g)=2SO2(g)+2H2O(l) ��H2=-1172kJ��mol-1
��H2S�����������Ӧ���ɹ�̬���Һ̬ˮ���Ȼ�ѧ����ʽΪ__________________��
��3��������K2CO3��Һ����H2S����ԭ��ΪK2CO3+H2S=KHS+KHCO3���÷�Ӧ��ƽ�ⳣ��Ϊ________��(��֪H2CO3��Ka1=4.2��10-7��Ka2=5.6��10-11��H2S��Ka1=5.6��10-8��Ka2=1.2��10-15)
��4����ǿ����Һ����H2O2�ɽ�COS����Ϊ��������һԭ��������COS�����÷�Ӧ��Ӧ�Ļ�ѧ����ʽΪ_________________��
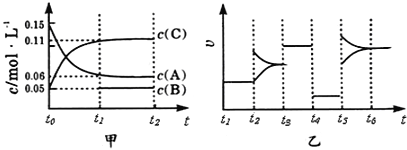
��5��COS��ˮ�ⷴӦΪCOS(g)+H2O(g)![]() CO2(g)+H2S(g) ��H<0��ij�¶�ʱ���û�����-Al2O3���������ں����ܱ�������COS(g)��ƽ��ת�����治ͬͶ�ϱ�[n(H2O)/n(COS)]��ת����ϵ��ͼ1��ʾ������������ͬʱ���ı䷴Ӧ�¶ȣ����һ��ʱ����COS��ˮ��ת������ͼ2��ʾ��
CO2(g)+H2S(g) ��H<0��ij�¶�ʱ���û�����-Al2O3���������ں����ܱ�������COS(g)��ƽ��ת�����治ͬͶ�ϱ�[n(H2O)/n(COS)]��ת����ϵ��ͼ1��ʾ������������ͬʱ���ı䷴Ӧ�¶ȣ����һ��ʱ����COS��ˮ��ת������ͼ2��ʾ��

���÷�Ӧ���������Ϊ:Ͷ�ϱ�[n(H2O)/n(COS)]____���¶�_____________
��P���Ӧ��ƽ�ⳣ��Ϊ_____________��(����С�����2λ)
�����¶����ߵ�һ��ֵ��������һ��ʱ����COS(g)��ˮ��ת���ʽ��ͣ��²���ܵ�ԭ����__________________��
���𰸡� ![]() 2H2S(g)+O2(g)=2S(s)+2H2O(l) ��H=-632kJ��mol-1 1.0��103 COS+4H2O2 =CO2+H2SO4+3H2O 10��1 160�� 0.05 �������Խ��ͣ�ƽ�����淴Ӧ�����ƶ�
2H2S(g)+O2(g)=2S(s)+2H2O(l) ��H=-632kJ��mol-1 1.0��103 COS+4H2O2 =CO2+H2SO4+3H2O 10��1 160�� 0.05 �������Խ��ͣ�ƽ�����淴Ӧ�����ƶ�
��������������������⿼�����ʽ����д����˹���ɵ�Ӧ�á�ƽ�ⳣ���ļ��㡢ָ���龳�·���ʽ����д����ѧƽ��ͼ�������
��1��COS�ĵ���ʽ��![]() ��
��
��2��Ӧ�ø�˹���ɣ�����![]() 2+�ڣ�
2+�ڣ�![]() 3����2H2S��g��+O2��g��=2S��s��+2H2O��l����H=[��-362kJ/mol��
3����2H2S��g��+O2��g��=2S��s��+2H2O��l����H=[��-362kJ/mol��![]() 2+��-1172kJ/mol��]
2+��-1172kJ/mol��]![]() 3=-632kJ/mol��H2S�����������Ӧ���ɹ�̬���Һ̬ˮ���Ȼ�ѧ����ʽΪ��2H2S��g��+O2��g��=2S��s��+2H2O��l����H=-632kJ/mol��
3=-632kJ/mol��H2S�����������Ӧ���ɹ�̬���Һ̬ˮ���Ȼ�ѧ����ʽΪ��2H2S��g��+O2��g��=2S��s��+2H2O��l����H=-632kJ/mol��
��3����ӦK2CO3+H2S=KHS+KHCO3�����ӷ���ʽΪCO32-+H2S=HS-+HCO3-���÷�Ӧ��ƽ�ⳣ��K=![]() =
=![]() =
=![]() =
=![]() =1
=1![]() 103��
103��
��4����ǿ����Һ��H2O2��COS����ΪH2SO4��ͬʱ����CO2��H2O2����ԭ���÷�Ӧ�Ļ�ѧ����ʽΪ4H2O2+COS=H2SO4+CO2+3H2O��
��5��������ͼ1������n��H2O��/n��COS��������COS��ƽ��ת��������n��H2O��/n��COS��![]() 10:1��COS��ƽ��ת����������������Ӧ��Ͷ�ϱ����Ϊn��H2O��/n��COS��=10:1������ͼ2��һ��ʱ����COS��ˮ��ת������160��ʱ���Ӧ������¶�Ϊ160�档
10:1��COS��ƽ��ת����������������Ӧ��Ͷ�ϱ����Ϊn��H2O��/n��COS��=10:1������ͼ2��һ��ʱ����COS��ˮ��ת������160��ʱ���Ӧ������¶�Ϊ160�档
��P��n��H2O��/n��COS��=6��ƽ��ʱCOS��ת����Ϊ40%������ʼc��COS��=amol/L������ʼc��H2O��=6amol/L��������ʽ
COS��g��+H2O��g��![]() CO2��g��+H2S��g��
CO2��g��+H2S��g��
c����ʼ����mol/L�� a 6a 0 0
c��ת������mol/L��0.4a 0.4a 0.4a 0.4a
c��ƽ�⣩��mol/L��0.6a 5.6a 0.4a 0.4a
P���Ӧ��ƽ�ⳣ��K=![]() =
=![]() =0.05��
=0.05��
�����¶����ߵ�һ��ֵ����һ��ʱ����COS��ˮ��ת���ʽ��ͣ�����ԭ���ǣ��������Խ��ͣ���Ӧ���ʱ������÷�Ӧ����ӦΪ���ȷ�Ӧ�������¶�ƽ�����淴Ӧ�����ƶ���

����Ŀ��һ���¶����������������Ϊ2.0L�ĺ����ܱ������зֱ����һ������X��������Ӧ��pX(g) ![]() Y(g)+Z(g)������������±���ʾ��
Y(g)+Z(g)������������±���ʾ��
������� | �¶�(��) | ��ʼ���ʵ���(mol) | ƽ�����ʵ���(mol) | |
X(g) | Y(g) | Z(g) | ||
�� | 387 | 0.20 | 0.080 | 0.080 |
�� | 387 | 0.40 | 0.160 | 0.160 |
�� | T | 0.20 | 0.090 | 0.090 |
�ش��������⣺
��1�����������з�Ӧ��10min�ﵽƽ�⣬��ǰ10min��Y��ƽ����Ӧ����v(Y)=___________��������������������ʼʱX�ķ�Ӧ����v(X)��___________v(X)��(������������С��������������)��
��2����֪������ӦΪ���ȷ�Ӧ����T___________387(��������������С����)���ж�������___________��
��3����Ӧ����ʽ��X�Ļ�ѧ������p��ȡֵΪ___________����������X��ƽ��ת����Ϊ___________������ʼʱ���������г���0.1molX��0.15molY��0.10molZ����Ӧ����___________ (����������������)��Ӧ����������ж�������_____________________________________________________��