��Ŀ����
��������ҵ���������ɡ���������ȡ������ұ�������ļӹ��Ȼ��ڹ��ɡ���ش��������⣺
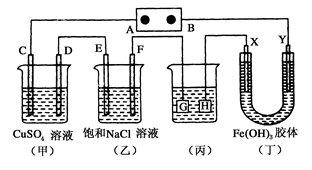
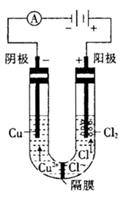
��1����ҵ�ϲ��õ���������ͱ���ʯ(Na3AlF6)������ķ���ұ���õ���������
2Al2O3 4Al��3O2��
4Al��3O2��
�������ʯ�����ã�________________________________________________��
��2��������������������������������Fe��Si�����ʣ����õ�ⷽ����һ���ᴿ���õ����������ĵ缫��ӦʽΪ________________�����п����������ϵ���__________��
A������ B��ʯī C��Ǧ�� D������
��3������������ʹ���������������ܵ�����Ĥ����ϡ����Ϊ���Һ�������������ĵ缫��ӦʽΪ_____________________________________________________________��
��4�������������������У���Ҫ���ϵص�����ѹ��������_________________��
��5������˵����ȷ����__________________��
A������������Ӧ��ԭ���ԭ�����н������ϱ��洦���ļ���
B������������������ǿ������ľ�Ե����
C������������������߽���������Ͻ����ʴ�ԣ�����ĥ���½�
D��������������Ĥ���ж���ԣ����к�ǿ���������ܣ�������Ⱦ�϶��ʸ�����ɫ
��1����ҵ�ϲ��õ���������ͱ���ʯ(Na3AlF6)������ķ���ұ���õ���������

2Al2O3
 4Al��3O2��
4Al��3O2���������ʯ�����ã�________________________________________________��
��2��������������������������������Fe��Si�����ʣ����õ�ⷽ����һ���ᴿ���õ����������ĵ缫��ӦʽΪ________________�����п����������ϵ���__________��
A������ B��ʯī C��Ǧ�� D������
��3������������ʹ���������������ܵ�����Ĥ����ϡ����Ϊ���Һ�������������ĵ缫��ӦʽΪ_____________________________________________________________��
��4�������������������У���Ҫ���ϵص�����ѹ��������_________________��
��5������˵����ȷ����__________________��
A������������Ӧ��ԭ���ԭ�����н������ϱ��洦���ļ���
B������������������ǿ������ľ�Ե����
C������������������߽���������Ͻ����ʴ�ԣ�����ĥ���½�
D��������������Ĥ���ж���ԣ����к�ǿ���������ܣ�������Ⱦ�϶��ʸ�����ɫ
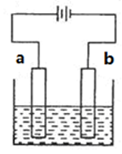
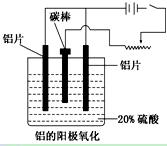
��1����Ҫ�ǽ���Al2O3���ۻ��¶�
��2��Al��3e��=Al3�� D
��3��2Al��6e����3H2O=Al2O3��6H��
��4�����������治�������������������Ϊ�˱����ȶ��ĵ�������Ҫ���������ѹ
��5��BD
��2��Al��3e��=Al3�� D
��3��2Al��6e����3H2O=Al2O3��6H��
��4�����������治�������������������Ϊ�˱����ȶ��ĵ�������Ҫ���������ѹ
��5��BD
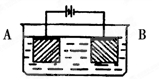
�����������1������Al2O3�۵�ܸߣ��������ʯ���Խ���Al2O3���۵㣬�����ۼ���
��2���ڵ�������������������Ӧ������ʧȥ���ӣ��缫��ӦʽΪAl��3e��=Al3������⾫����Ӧ�Դ������������ϣ���D����ȷ��
��3��������������������Ӧ���Եõ��缫��ӦʽΪ��2Al��3H2O��6e��=Al2O3��6H����
��4�����������������ﱡĤ���ʹ��������Ϊ�˱����ȶ��ĵ�������Ҫ�������������������ѹ��
��5����������Ӧ�õ��ǵ��ԭ����A�������������������ɿ�����ǿ������ľ�Ե�ԣ�B����ȷ����������������������������Ͻ����ʴ�ԣ�����ĥ�Բ��½���C��������������в�ͬ�ľ��ͣ����еĦ�-Al2O3���ж���ԡ�ǿ�����Ժʹ����ԣ������������ʹ�����D����ȷ��

��ϰ��ϵ�д�
�����Ŀ