��Ŀ����
�ڵ�ˮ�����У���;��ҵ��ˮ�������ŷ������ˮ�ʶ�������������ij������Һ�У����д���Mg2+��Al3+��Cu2+��Ag+���Է����ش��������⣺
��1���÷�Һ�п��ܴ������ڵ�һ����������
A��SO42-������B��NO3-C��Cl-������D��CO32-
��2��Ϊ����Һ����Ԫ�صĺ������轫��ӷ�ˮ��Ʒ�з�����������õ��Լ���
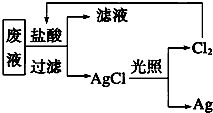
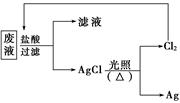
��3��Ϊ�˻��շ�Һ�еĽ�������ijͬѧ�������ͼ��ʾ������
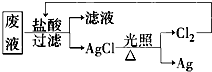
�����÷������108g Ag��Ϊ��֤����Ⱦ������Cl2��ѭ�����ã�������Ӧ�ṩ��״����H2�������
��1���÷�Һ�п��ܴ������ڵ�һ����������
B
B
������ţ�A��SO42-������B��NO3-C��Cl-������D��CO32-
��2��Ϊ����Һ����Ԫ�صĺ������轫��ӷ�ˮ��Ʒ�з�����������õ��Լ���
NaOH��Һ
NaOH��Һ
����Ԫ�ط����仯�����ӷ���ʽ��Al3++4OH-�TAlO2-+2H2O
Al3++4OH-�TAlO2-+2H2O
����3��Ϊ�˻��շ�Һ�еĽ�������ijͬѧ�������ͼ��ʾ������

�����÷������108g Ag��Ϊ��֤����Ⱦ������Cl2��ѭ�����ã�������Ӧ�ṩ��״����H2�������
11.2L
11.2L
����������1������������Һˮ���Ȼ���������ˮ������������̼����������ɲ�����ˮ��̼���Σ�
��2��ֻ������������Ӧ�����ü���������ӷ��룻
��3����2Ag��Cl2��H2�����㣮
��2��ֻ������������Ӧ�����ü���������ӷ��룻
��3����2Ag��Cl2��H2�����㣮
����⣺��1��������������Һˮ���Ȼ���������ˮ������������̼����������ɲ�����ˮ��̼���Σ����е������ζ�����ˮ���������������������Ӿ�����Ӧ���ʴ�Ϊ��B����
��2��ֻ������������Ӧ�����ü���������ӷ��룬���Լ�ѡNaOH��Һ�����������ӷ�ӦΪAl3++4OH-�TAlO2-+2H2O��
�ʴ�Ϊ��NaOH��Һ��Al3++4OH-�TAlO2-+2H2O����
��3��n��Ag��=
=1mol����2Ag��Cl2��H2��֪��������Ӧ�ṩ��״����H2�����Ϊ0.5mol��22.4L/mol=11.2L���ʴ�Ϊ��11.2L��
��2��ֻ������������Ӧ�����ü���������ӷ��룬���Լ�ѡNaOH��Һ�����������ӷ�ӦΪAl3++4OH-�TAlO2-+2H2O��
�ʴ�Ϊ��NaOH��Һ��Al3++4OH-�TAlO2-+2H2O����
��3��n��Ag��=
| 108g |
| 108g/mol |
���������⿼�����ӵĹ��棬���ӵķ��뼰�йؼ��㣬��Ϥ����֮��ķ�Ӧ�����ӵ����ʼ��ɽ����Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
 ����Ժǿ��������ˮ���������̱����ֽ�ˮ�����ۺ���̬���������빤�̽�����Э������ˮ��Դ��������Ϊ���ߣ��ѽ�ˮ�����ۡ���̬�����������ˮ��Ϊһ��������ϵͳ��������ԭ���ڵ�ˮ�����У���;��ҵ��ˮ�������ŷ������ˮ�ʶ�������������ij������Һ�У����д�����Mg2+��Al3+��Cu2+��Ag+���Է����ش��������⣺
����Ժǿ��������ˮ���������̱����ֽ�ˮ�����ۺ���̬���������빤�̽�����Э������ˮ��Դ��������Ϊ���ߣ��ѽ�ˮ�����ۡ���̬�����������ˮ��Ϊһ��������ϵͳ��������ԭ���ڵ�ˮ�����У���;��ҵ��ˮ�������ŷ������ˮ�ʶ�������������ij������Һ�У����д�����Mg2+��Al3+��Cu2+��Ag+���Է����ش��������⣺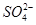 B��
B��
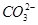
 4H����2Fe2����4SO42����
4H����2Fe2����4SO42����