��Ŀ����
 ��50mL0.50mol/L������50mL0.55mol/LNaOH��Һ����ͼ��ʾ��װ���н����кͷ�Ӧ��ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ��ش��������⣺
��50mL0.50mol/L������50mL0.55mol/LNaOH��Һ����ͼ��ʾ��װ���н����кͷ�Ӧ��ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ��ش��������⣺��1����ʵ��װ���Ͽ���ͼ����ȱ�ٵ�һ�ֲ���������
����������
����������
����2���ձ���������ֽ����������
����ʵ������е�������ʧ
����ʵ������е�������ʧ
����3��Ҫ�ظ���������ʵ���Ŀ����
��β�����ƽ��ֵ���Լ���ʵ�����
��β�����ƽ��ֵ���Լ���ʵ�����
����4�����ձ����粻��Ӳֽ�壬��õ��к�����ֵ
ƫС
ƫС
���ƫ��ƫС����Ӱ�족���������µ���10��ʱ���У���ʵ��������ɽϴ������ԭ�������½ϵ�ʱ��Ӧ��ϵ��ɢ�ȱȽϿ죬������ʧ���ƫ��
���½ϵ�ʱ��Ӧ��ϵ��ɢ�ȱȽϿ죬������ʧ���ƫ��
��5�������60mL0.50mol/L������50mL0.55mol/LNaOH��Һ���з�Ӧ��������ʵ����ȣ����ų�������
�����
�����
�����ȡ�����ȡ����������к������
���
�����ȡ�����ȡ���������������Ϊ�к�����ָ�������кͷ�Ӧ����nmolH2O���ų���������������������
��Ϊ�к�����ָ�������кͷ�Ӧ����nmolH2O���ų���������������������
����6������ͬŨ�Ⱥ�����Ĵ��ᣨCH3COOH������HCl��Һ��������ʵ�飬��õ��к��ȵ���ֵ��
ƫС
ƫС
�����ƫ��ƫС����Ӱ�족������7������ƽ�в�������õ��������£�
| �¶� ��� |
��ʼ�¶�t1/�� | ��ֹ�¶� T2/�� |
�¶Ȳ� ��t/�� | ||
| HCl | NaOH | ƽ��ֵ | |||
| 1 | 25 | 25 | 27.3 | ||
| 2 | 25 | 25 | 27.4 | ||
| 3 | 25 | 25 | 28.6 | ||
46.3kJ?mol-1
46.3kJ?mol-1
����������1���������ȼƵĹ������жϸ�װ�õ�ȱ��������
��2���к��Ȳⶨʵ��ɰܵĹؼ��DZ��¹�����
��3����β�������ʵ����
��4������Ӳֽ�壬����һ��������ɢʧ��
��5����Ӧ�ų����������������Լ�������Ķ����йأ��������к��ȵĸ����ʵ�����ش�
��6������������ʵ������ȷ�����
��7�����ݷ�Ӧ����Һ�ı�����cΪ4.18J?��-1?g-1�������ʵ��ܶȾ�Ϊ1g?cm-3�� ���빫ʽ��Q=cm��t2-t1�������㼴�ɣ�
��2���к��Ȳⶨʵ��ɰܵĹؼ��DZ��¹�����
��3����β�������ʵ����
��4������Ӳֽ�壬����һ��������ɢʧ��
��5����Ӧ�ų����������������Լ�������Ķ����йأ��������к��ȵĸ����ʵ�����ش�
��6������������ʵ������ȷ�����
��7�����ݷ�Ӧ����Һ�ı�����cΪ4.18J?��-1?g-1�������ʵ��ܶȾ�Ϊ1g?cm-3�� ���빫ʽ��Q=cm��t2-t1�������㼴�ɣ�
����⣺��1���������ȼƵĹ����֪��װ�õ�ȱ�������ǻ��β������������ʴ�Ϊ�����β�����������
��2���к��Ȳⶨʵ��ɰܵĹؼ��DZ��¹�������С�ձ�֮��������ֽ���������ǣ�����ʵ������е�������ʧ���ʴ�Ϊ������ʵ������е�������ʧ��
��3����β�������ʵ�����ʴ�Ϊ����β�����ƽ��ֵ���Լ���ʵ����
��4�����ձ����粻��Ӳֽ�壬����һ��������ɢʧ����õ��к�����ֵ�����С���ʴ�Ϊ��ƫС�����½ϵ�ʱ��Ӧ��ϵ��ɢ�ȱȽϿ죬������ʧ���ƫ�ͣ�
��5����Ӧ�ų����������������Լ��������7���йأ�������60mL0.25mol?L-nH2SO4��Һ��50mL0.55mol?L-nNaOH��Һ���з�Ӧ��������ʵ����ȣ�����ˮ������7�����ų�������ƫ�ߣ������к��ȵľ���ǿ���ǿ�Ӧ����nmolˮʱ�ų����ȣ������������أ�������50mL0.50mol?L-n�������H2SO4��Һ��������ʵ�飬����к�����ֵ��ȣ��ʴ�Ϊ������ȣ���ȣ���Ϊ�к�����ָ�������кͷ�Ӧ����nmolH2O���ų��������������������أ�
��6������Ϊ���ᣬ�������Ϊ���ȹ��̣����Դ��ᣨCH3COOH������HCl��Һ��Ӧ����Ӧ�ų�������С��57��jkJ���ʴ�Ϊ��ƫС��
��7����t=
=2.77��Q=cm��t��2.77��C��4.18J/��g?�棩��100g=1159J=1.159kJ����H=-Q/n��H2O��=-1.159kJ/0.025mol=-46.3kJ?mol-1���ʴ�Ϊ��46.3kJ?mol-1��
��2���к��Ȳⶨʵ��ɰܵĹؼ��DZ��¹�������С�ձ�֮��������ֽ���������ǣ�����ʵ������е�������ʧ���ʴ�Ϊ������ʵ������е�������ʧ��
��3����β�������ʵ�����ʴ�Ϊ����β�����ƽ��ֵ���Լ���ʵ����
��4�����ձ����粻��Ӳֽ�壬����һ��������ɢʧ����õ��к�����ֵ�����С���ʴ�Ϊ��ƫС�����½ϵ�ʱ��Ӧ��ϵ��ɢ�ȱȽϿ죬������ʧ���ƫ�ͣ�
��5����Ӧ�ų����������������Լ��������7���йأ�������60mL0.25mol?L-nH2SO4��Һ��50mL0.55mol?L-nNaOH��Һ���з�Ӧ��������ʵ����ȣ�����ˮ������7�����ų�������ƫ�ߣ������к��ȵľ���ǿ���ǿ�Ӧ����nmolˮʱ�ų����ȣ������������أ�������50mL0.50mol?L-n�������H2SO4��Һ��������ʵ�飬����к�����ֵ��ȣ��ʴ�Ϊ������ȣ���ȣ���Ϊ�к�����ָ�������кͷ�Ӧ����nmolH2O���ų��������������������أ�
��6������Ϊ���ᣬ�������Ϊ���ȹ��̣����Դ��ᣨCH3COOH������HCl��Һ��Ӧ����Ӧ�ų�������С��57��jkJ���ʴ�Ϊ��ƫС��
��7����t=
| 27.3+27.4+28.6-25��3 |
| 3 |
���������⿼���Ȼ�ѧ����ʽ�Լ���Ӧ�ȵļ��㣬��Ŀ�Ѷȴ�ע�������к��ȵĸ����Լ��ⶨ��Ӧ�ȵ��������⣮

��ϰ��ϵ�д�
�����Ŀ
 ��50mL0.50mol/L������50mL0.55mol/LNaOH��Һ����ͼ��ʾ��װ���н����кͷ�Ӧ��ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ��ش��������⣺
��50mL0.50mol/L������50mL0.55mol/LNaOH��Һ����ͼ��ʾ��װ���н����кͷ�Ӧ��ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ��ش��������⣺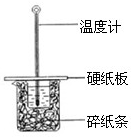 ��1�����ⶨ��20g������������ȼ������ˮ����������2418.0kJ��д���÷�Ӧ���Ȼ�ѧ����ʽ
��1�����ⶨ��20g������������ȼ������ˮ����������2418.0kJ��д���÷�Ӧ���Ȼ�ѧ����ʽ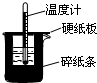 50mL0.50mol/L������50mL0.55mol/LNaOH��Һ����ͼ��ʾ��װ���н����кͷ�Ӧ��ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ��ش��������⣺
50mL0.50mol/L������50mL0.55mol/LNaOH��Һ����ͼ��ʾ��װ���н����кͷ�Ӧ��ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ��ش��������⣺