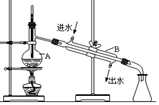
��Ŀ����
���÷Ͼɶ�п��Ƥ���Ʊ�����Fe3O4�������Ӽ�������ZnO���Ʊ��������£�
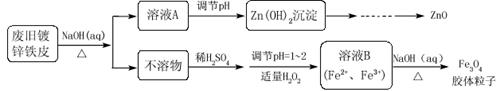
��֪��Zn���仯�����������Al���仯������������ơ���ش��������⣺
��1����NaOH��Һ�����Ͼɶ�п��Ƥ����ȥ�����ۣ�������____________________��
��2��������ҺA��pH�ɲ���Zn(OH)2������Ϊ�Ƶ�ZnO����������������_____��ϴ�ӡ�_____��
��3������ҺB�Ƶ�Fe3O4�������ӵĹ����У������ͨ��N2����ԭ����________________��
��4�����ظ���ط���һ��������ԭ�ζ������ɲⶨ����Fe3O4�еĶ�������������д������������K2Cr2O7��Fe2+��Ӧ�����ӷ���ʽ ��K2Cr2O7����ԭΪCr3+����
��5����������Ũ��Ϊ0.01000 mol·L-1��K2Cr2O7����Һ250mL��Ӧȷ��ȡK2Cr2O7 g��������λ��Ч���֣���֪M(K2Cr2O7)="294.0" g·mol-1�������Ƹñ���Һʱ����������һ����Ҫ�õ����� ���ñ�ű�ʾ����
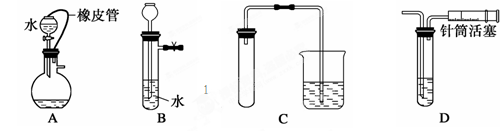
�ٵ�����ƽ ���ձ� ����Ͳ �ܲ����� ��250 mL����ƿ ��ͷ�ι� ��������ƽ
��6��������K2Cr2O7����Һʱ�����ӿ̶��ߣ���ⶨ���_______���ƫ����ƫС�����䡱����ͬ�����ζ������У����ζ�ǰװ��K2Cr2O7����Һ�ĵζ��ܼ��첿�������ݣ����ζ�������������ʧ����ζ������________��

��֪��Zn���仯�����������Al���仯������������ơ���ش��������⣺
��1����NaOH��Һ�����Ͼɶ�п��Ƥ����ȥ�����ۣ�������____________________��
��2��������ҺA��pH�ɲ���Zn(OH)2������Ϊ�Ƶ�ZnO����������������_____��ϴ�ӡ�_____��
��3������ҺB�Ƶ�Fe3O4�������ӵĹ����У������ͨ��N2����ԭ����________________��
��4�����ظ���ط���һ��������ԭ�ζ������ɲⶨ����Fe3O4�еĶ�������������д������������K2Cr2O7��Fe2+��Ӧ�����ӷ���ʽ ��K2Cr2O7����ԭΪCr3+����
��5����������Ũ��Ϊ0.01000 mol·L-1��K2Cr2O7����Һ250mL��Ӧȷ��ȡK2Cr2O7 g��������λ��Ч���֣���֪M(K2Cr2O7)="294.0" g·mol-1�������Ƹñ���Һʱ����������һ����Ҫ�õ����� ���ñ�ű�ʾ����
�ٵ�����ƽ ���ձ� ����Ͳ �ܲ����� ��250 mL����ƿ ��ͷ�ι� ��������ƽ
��6��������K2Cr2O7����Һʱ�����ӿ̶��ߣ���ⶨ���_______���ƫ����ƫС�����䡱����ͬ�����ζ������У����ζ�ǰװ��K2Cr2O7����Һ�ĵζ��ܼ��첿�������ݣ����ζ�������������ʧ����ζ������________��
��1���ܽ��п�� ��2�����ˡ����� ��3��N2�����£���ֹFe2+������
��4��6Fe2+ + Cr2O72- + 14H+ = 6Fe3+ + 2Cr3+ + 7H2O ��5��0.7350 �ۢ� ��6��ƫС ƫ��
��4��6Fe2+ + Cr2O72- + 14H+ = 6Fe3+ + 2Cr3+ + 7H2O ��5��0.7350 �ۢ� ��6��ƫС ƫ��
�����������1��NaOH��Һ����п������Ӧ��������NaOH��Һ�����Ͼɶ�п��Ƥ����ȥ�����ۣ��������ܽ��п�㡣��2����Ӧ����Zn(OH)2���������ȷֽ��������п��ˮ������Ϊ�Ƶ�ZnO���������������ǹ��ˡ�ϴ�ӡ����ա���3��������ѧ���ʲ����ã������ֶ��Ի����пɷ�ֹFe2+����������4����Ӧ�����ַ���ʽ�ǣ�6Fe2+ + Cr2O72- + 14H+ = 6Fe3+ + 2Cr3+ + 7H2O ��5��m=n·M="0.01mol/L��0.25L��294.0" g/mol=0.7350g.�������ʵ���Ũ�ȵ���Һ�õ��������Тٵ�����ƽ ���ձ� ����Ͳ �ܲ����� ��250 mL����ƿ ��ͷ�ιܡ���6��������K2Cr2O7����Һʱ�����ӿ̶��ߣ�������Һ�����ƫС����K2Cr2O7����ҺŨ��ƫ�������ֱ���Һ�ⶨ�����ĵı���Һ��ƫС���ƫС���ζ������У����ζ�ǰװ��K2Cr2O7����Һ�ĵζ��ܼ��첿�������ݣ����ζ�������������ʧ�����ĵı���Һ���������ƫ����ζ������ƫ��

��ϰ��ϵ�д�
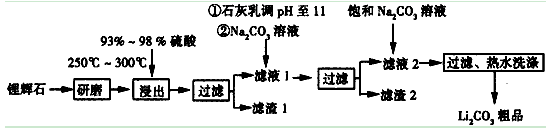
�����Ŀ
 H2SO4(Ũ)
H2SO4(Ũ) Li2SO4
Li2SO4 (Li2CO3)/g
(Li2CO3)/g