��Ŀ����
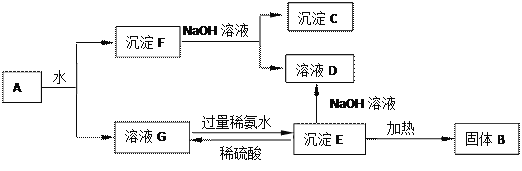
����Ŀ��ij��Һ�п��ܺ���H+��Mg2+��NH4+��Al3+��Fe3+��CO32����SO42����NO3���еļ��֡�������п��,������ɫ��ζ������;������NaOH��Һ,������ɫ����,�Ҳ����ij����������NaOH�����ʵ���֮��Ĺ�ϵ��ͼ��ʾ��������˵������ȷ����

A. ��Һ�е���������H+��Mg2+��NH4+��Al3+
B. ��Һ��n(Al3+)=0.1 mol
C. ��Һ��һ������CO32��,���ܺ���SO42����NO3��
D. n(H+)��n(Al3+)��n(Mg2+)=2��2��1
���𰸡�C
��������
��п������ɫ��ζ����˵��п�������ӷ�Ӧ������������Һ���д��������ӣ���û��̼�������������ӣ�������������Һ������ɫ����˵�����������ӣ���ͼ���֪��Һ�к������ӡ�笠����ӡ�þ���ӡ������ӣ���������������Һ��������������þ���Ӻ���������笠���������������������˳��Ӧ���������ı仯������x���ϣ�Ȼ��������ƽ��x�ᣬ�ټ�С�����ƽ��x�ᣬ���Զ���ͼ���ϵ�����������ӵ�����
��ͼ���֪��Һ�к������ӡ�笠����ӡ�þ���ӡ������ӣ�A��ȷ���ܽ��������������������Ƶ����ʵ���Ϊ��0.8mol��0.7mol��=0.1mol�����������������ܽ�ķ���ʽ��֪���������������ʵ���Ϊ0.1mol������ԭ�Ӹ����غ��֪�����ӵ����ʵ���Ϊ0.1mol��B��ȷ����п������ɫ��ζ����˵��п�������ӷ�Ӧ������������Һ���д��������ӣ���û��̼�������������ӣ�C������ͼ�����ݿ�֪���к������ӵ��������Ƶ����ʵ���Ϊ0.1mol����Һ�������ӵ����ʵ���Ϊ0.1mol���ܽ����������������������Ƶ����ʵ���Ϊ0.1mol���������������ʵ���Ϊ0.1mol������ԭ�Ӹ����غ��֪�����ӵ����ʵ���Ϊ0.1mol������������þ�������������������������Ƶ����ʵ���Ϊ��0.5mol��0.1mol��=0.4mol�������������������������Ƶ����ʵ���Ϊ0.3mol��������������þ���������������Ƶ����ʵ���Ϊ0.1mol���ɷ���ʽ��֪������þ�����ʵ���Ϊ0.05mol����þԭ�Ӹ����غ��֪þ���ӵ����ʵ���Ϊ0.05mol���������ӡ������Ӻ�þ���ӵ����ʵ�����Ϊ2:2:1��D��ȷ��
��ѡC��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
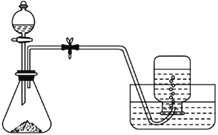
Сѧ��10����Ӧ����ϵ�д�����Ŀ����֪���ᾧ��(H2C2O4��XH2O)������ˮ�����������Ը��������Һ��ȫ��Ӧ:2KMnO4+5H2C2O4+3H2SO4==K2SO4+2MnSO4+10CO2��+8H2O������������ԭ�ζ����ⶨ���ᾧ��Ľᾧˮ������X����������:
���÷�����ƽ��ȡ���ᾧ��1.260g���������Ƴ�100.00mL���������Һ
������Һ����ȡ25.00mL���������Һ����ƿ�У����������������ữ
����Ũ��Ϊ0.1000mol/L��KMnO4����Һ���еζ������ν������:
��һ�εζ� | �ڶ��εζ� | �����εζ� | |
������Һ���(mL) | 25.00 | 25.00 | 25.00 |
����Һ���(mL) | 9.99 | 10.01 | 10.00 |
��֪H2C2O4����Է�������Ϊ90����ش���������:
��1���ζ�ʱ��KMnO4����ҺӦ��װ��________(����ʽ���ʽ)�ζ����С�
��2��������ʵ�������,����Ҫ����������Ʒ��______(�����)��
��100mL����ƿ���ձ��۵ζ��ܼТ�©���ݲ�������������ƽ
��3������ζ��յ�ı�־��____________________________________________��
��4�������������ݼ���X=________________��
��5��������(��ƫ�ߡ�ƫ�ͻ���Ӱ��):
�����ζ���ʼʱ���ӵζ��̶ܿȣ��ζ�����ʱ���ӵζ��̶ܿȣ���Xֵ________��
����KMnO4����ҺŨ��ƫ�ͣ���Xֵ_________��
������ƿϴ����ƿ�ڻ���������������ˮ����Xֵ____________��