ЬтФПФкШн
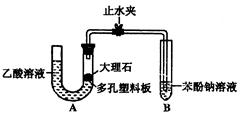
(13 Зж)гавЛАќЙЬЬхЗлФЉЃЌгЩCaCO3ЁЂNa2SO4ЁЂKClЁЂBa (NO3)2ЁЂCuSO4жаЕФШ§жжЮяжЪзщГЩЃЌШЁбљЦЗНјааШчЯТЪЕбщЃК

ЃЈ1ЃЉаДГіЪЕбщЙ§ГЬжаЗЂЩњЗДгІЕФЛЏбЇЗНГЬЪНЃК ЁЃ
ЃЈ2ЃЉЙЬЬхЗлФЉжавЛЖЈВЛДцдкЕФЮяжЪЪЧ ЃЛвЛЖЈДцдкЕФЮяжЪЪЧ ЃЛ
ЃЈ3ЃЉНЋЙЬЬхЗлФЉПЩФмЕФзщКЯЬюШыЯТБэЃКЃЈПЩвдВЛЬюТњЃЌвВПЩвддйВЙГфЁЃЃЉ
ЃЈ4ЃЉЧыЩшМЦвЛИіЪЕбщНјвЛВНШЗЖЈЛьКЯЮяЕФзщГЩЃЌМђЪіЪЕбщВНжшЁЂЯжЯѓКЭНсТлЁЃ

ЃЈ1ЃЉаДГіЪЕбщЙ§ГЬжаЗЂЩњЗДгІЕФЛЏбЇЗНГЬЪНЃК ЁЃ
ЃЈ2ЃЉЙЬЬхЗлФЉжавЛЖЈВЛДцдкЕФЮяжЪЪЧ ЃЛвЛЖЈДцдкЕФЮяжЪЪЧ ЃЛ
ЃЈ3ЃЉНЋЙЬЬхЗлФЉПЩФмЕФзщКЯЬюШыЯТБэЃКЃЈПЩвдВЛЬюТњЃЌвВПЩвддйВЙГфЁЃЃЉ
| ађКХ | ЛЏбЇЪН |
| Ђё | |
| Ђђ | |
| Ђѓ | |
ЃЈ1ЃЉCaCO3+2HCl=CaCl2+H2O+CO2ЁќЃЈ2ЗжЃЉ
ЃЈ2ЃЉCuSO4ЃЛЃЈ2ЗжЃЉ CaCO3ЁЂKClЃЛЃЈ2ЗжЃЉ
ЃЈ3ЃЉЃЈИї2ЗжЃЉ
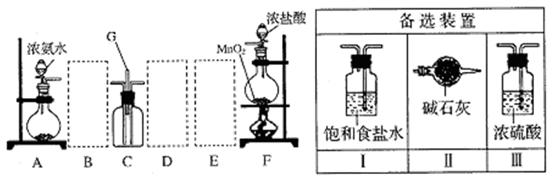
ЃЈ4ЃЉгУЪдЙмШЁЪЪСПЂйЙ§ТЫЕУЕФЮоЩЋШмвКЃЌМгШыЩйСПBaCl2ШмвКЃЌШєВњЩњАзЩЋГСЕэЃЌдђЮЊзщКЯIЃЌЗёдђЮЊзщКЯIIЁЃЃЈ3ЗжЃЉ
ЃЈ2ЃЉCuSO4ЃЛЃЈ2ЗжЃЉ CaCO3ЁЂKClЃЛЃЈ2ЗжЃЉ
ЃЈ3ЃЉЃЈИї2ЗжЃЉ
| I | CaCO3ЁЂNa2SO4ЁЂKCl |
| II | CaCO3ЁЂKClЁЂBa (NO3)2 |
Тд

СЗЯАВсЯЕСаД№АИ
ЯрЙиЬтФП



 ЪЧвЛжжДѓЦјЮлШОЮяЃЌФГаЫШЄаЁзщгћЬНОП
ЪЧвЛжжДѓЦјЮлШОЮяЃЌФГаЫШЄаЁзщгћЬНОП


 ЂёЃЎВщдФзЪСЯЃКЂйCu2OЪєгкМюадбѕЛЏЮяЃЛ ЂкдкПеЦјжазЦЩеCu2OЩњГЩCuOЃЛЂлCu2OдкЫсадЬѕМўЯТФмЗЂЩњЯТСаЗДгІЃКCu2OЃЋ2H+ЃНCuЃЋCu2+ЃЋH2OЁЃ
ЂёЃЎВщдФзЪСЯЃКЂйCu2OЪєгкМюадбѕЛЏЮяЃЛ ЂкдкПеЦјжазЦЩеCu2OЩњГЩCuOЃЛЂлCu2OдкЫсадЬѕМўЯТФмЗЂЩњЯТСаЗДгІЃКCu2OЃЋ2H+ЃНCuЃЋCu2+ЃЋH2OЁЃ бщЗНАИЃК
бщЗНАИЃК
 (НќЫЦжЕ)ЕФЪЕбщЁЃЯШГЦСПЗДгІЮябѕЛЏЭЕФжЪСПm(CuO)ЃЌЗДгІЭъШЋКѓВтЖЈЩњГЩЮяЫЎЕФжЪСП
(НќЫЦжЕ)ЕФЪЕбщЁЃЯШГЦСПЗДгІЮябѕЛЏЭЕФжЪСПm(CuO)ЃЌЗДгІЭъШЋКѓВтЖЈЩњГЩЮяЫЎЕФжЪСП m(H20)ЃЌгЩДЫМЦЫу
m(H20)ЃЌгЩДЫМЦЫу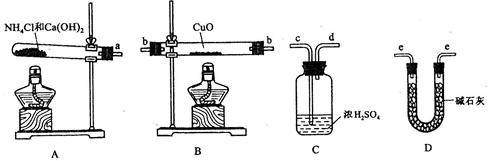
 ЕФЪЧ_______________ (ЬюађКХ)ЃЛ
ЕФЪЧ_______________ (ЬюађКХ)ЃЛ ИЩдя
ИЩдя _____КЭ_______________ДяЕНЪЕбщФПЕФЁЃ
_____КЭ_______________ДяЕНЪЕбщФПЕФЁЃ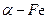 ЗлОпгаГЌЧПЕФДХадФмЃЌПЩгУзїИпУмЖШДХМЧТМЕФНщжЪвдМАИпаЇДпЛЏМСЕШЁЃдкВЛЭЌЮТЖШЯТЃЌ
ЗлОпгаГЌЧПЕФДХадФмЃЌПЩгУзїИпУмЖШДХМЧТМЕФНщжЪвдМАИпаЇДпЛЏМСЕШЁЃдкВЛЭЌЮТЖШЯТЃЌ
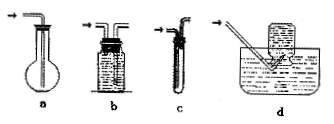
 ЪЧ ЃЈ ЃЉ
ЪЧ ЃЈ ЃЉ СђЫсЪБЃЌгІНЋХЈСђЫсЛКЛКзЂШыЪЂгаЫЎЕФСПЭВжа
СђЫсЪБЃЌгІНЋХЈСђЫсЛКЛКзЂШыЪЂгаЫЎЕФСПЭВжа