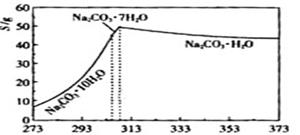
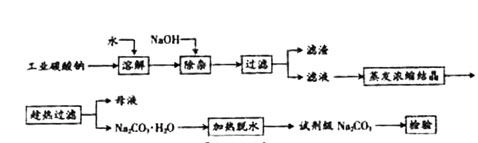
��Ŀ����
������������ʵ����ѧ2009������꼶�������¿�����ѧ��8��ijѧ������ʵ��������з�����ϴ����������
����Ũ��ˮ��ϴ����������Ӧ����Թ� ���þƾ���ϴ�������������ձ� ����Ũ������ϴ����������طֽ�ʵ����Թ� ����������ϴ���ڴ�����Ȼ�����Һ���Լ�ƿ ��������������Һ��ϴʢ�����ӵ��Թ� �������������Ƶ�Ũ��Һ��ϴմ����ǵ��Թ�
���ж����ϲ������жϡ���ȷ����
����Ũ��ˮ��ϴ����������Ӧ����Թ� ���þƾ���ϴ�������������ձ� ����Ũ������ϴ����������طֽ�ʵ����Թ� ����������ϴ���ڴ�����Ȼ�����Һ���Լ�ƿ ��������������Һ��ϴʢ�����ӵ��Թ� �������������Ƶ�Ũ��Һ��ϴմ����ǵ��Թ�
���ж����ϲ������жϡ���ȷ����
| A�������ⶼ�� | B�������ⶼ�� | C���ܢݲ��� | D��ȫ����ȷ |
A
�ٲ���ȷ������������Ӧ����Թ�Ӧ��ϡ������ϴ�������ȷ,ԭ��������ʽΪ���ڵ������ھƾ�����MnO2��4HCl=MnCl2��Cl2����2H2O��Fe(OH)3��3HCl=FeCl3��3H2O��NaOH��C6H5OH��C6H5ONa��H2O��3S��6NOH=2Na2S��Na2SO3��3H2O

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ

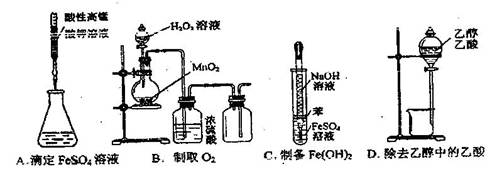
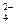 �����ʣ��ᴿ����·�����£�
�����ʣ��ᴿ����·�����£�