��Ŀ����
2��ij�ᾧˮ����Ļ�ѧʽΪR•nH2O������Է�������ΪM.25��ʱ��ag�þ����ܹ�����bgˮ���γ�VmL��Һ�����й�ϵ�в���ȷ���ǣ�������| A�� | ����Һ�����ʵ���������Ϊ��=$\frac{100a��M-18n��}{a+b}$% | |
| B�� | ����Һ�����ʵ���Ũ��Ϊc=$\frac{1000a}{MV}$mol•L-1 | |
| C�� | ����Һ���ܼ������ʵ�������Ϊm��ˮ����m�����ʣ�=��$\frac{18na}{M}+b$������a-$\frac{18na}{M}$�� | |
| D�� | ����Һ���ܶ�Ϊ��=$\frac{1000��a+b��}{V}$g•L-1 |
���� A�����ݻ�ѧʽ����R����������Һ������Ϊ��a+b��g������w�����ʣ�=$\frac{m�����ʣ�}{m����Һ��}$��100%�������Һ����������
B������n=$\frac{m}{M}$����R•nH2O�����ʵ�������n��R��=n��R•nH2O��������c=$\frac{n}{V}$�������Һ�����ʵ���Ũ�ȣ�
C�����ݻ�ѧʽ����ᾧˮ������������R������Ϊ��ag-�ᾧˮ����������Һ���ܼ�����Ϊ���ᾧˮ����+bg����
D������c=$\frac{n}{V}$����ñ�����Һ�����ʵ���Ũ�ȣ����ݦ�=$\frac{m}{V}$������Һ�ܶȣ�������c=$\frac{1000�Ѧ�}{M}$���й�ʽ���μ��㣮
��� �⣺A��R������Ϊ$\frac{M-18n}{M}$��ag����Һ������Ϊ��a+b��g����֪����Һ��������Ϊ$\frac{\frac{M-18n}{M}��ag}{��a+b��g}$��100%=$\frac{100a��M-18n��}{M��a+b��}$%����A����
B��n��R��=n��R•nH2O��=$\frac{a}{M}$mol������Һ�����ʵ���Ũ��$\frac{\frac{a}{M}mol}{V��1{0}^{-3}L}$=$\frac{1000a}{MV}$mol/L����B��ȷ��
C��R•nH2O�нᾧˮ������Ϊ$\frac{18na}{M}$g����R������Ϊ��a-$\frac{18na}{M}$��g����Һ���ܼ�������Ϊ��$\frac{18na}{M}+b$��g������Һ��m��ˮ����m�����ʣ�=��$\frac{18na}{M}+b$������a-$\frac{18na}{M}$������C��ȷ��
D����Һ������Ϊ��a+b��g�����ݦ�=$\frac{m}{V}$��֪����Һ�ܶ�Ϊ$\frac{��a+b��g}{VmL}$=$\frac{a+b}{V}$g/mL=$\frac{1000��a+b��}{V}$g/L����D��ȷ��
��ѡA��
���� ���⿼����ҺŨ�ȼ��㣬�漰���ʵ���Ũ�ȡ�����������������ĸ�ͼ��㣬Ϊ�״���Ŀ��ע��Ի���֪ʶ���������գ�

 �Űٷֿ�ʱ����ϵ�д�
�Űٷֿ�ʱ����ϵ�д�| A�� | ���е���֬������ʹ��ˮ��ɫ | |
| B�� | ���Ͳ�˿����Ҫ�ɷݶ�����ά�� | |
| C�� | ���ǡ����ۡ���֬�ȶ����Է���ˮ�ⷴӦ | |
| D�� | ʳ����ά�ء����ۡ������ʡ�ֲ���Ͷ��Ǹ߷��ӻ����� |
| A�� | ֻ��Na2O2 | B�� | ֻ��Na2O | C�� | Na2O��Na2O2 | D�� | ��ȷ�� |
| A�� | ��ϩ | B�� | ��Ȳ | C�� | ��Ȳ | D�� | ��Ȳ |
 2014��10�£��������������ȶ���������г���������������������ѧУ����ͣ�Σ���������ġ������֮һ������β������������β��������Ҫ����Ԫ��������������Ҫ��Ⱦ�CO��NOx��̼�⻯������Է������Ӧ��������壬���ٻ�����Ⱦ���䷴Ӧԭ��֮һ��2NO��g��+2CO��g���TN2��g��+2CO��g����H���ش��������⣺
2014��10�£��������������ȶ���������г���������������������ѧУ����ͣ�Σ���������ġ������֮һ������β������������β��������Ҫ����Ԫ��������������Ҫ��Ⱦ�CO��NOx��̼�⻯������Է������Ӧ��������壬���ٻ�����Ⱦ���䷴Ӧԭ��֮һ��2NO��g��+2CO��g���TN2��g��+2CO��g����H���ش��������⣺��1������֪���з�Ӧ��
��N2��g��+O2��g���T2NO��g����H1=+180.8kJ/mol
��2C��s��+O2��g���T2CO��g����H2=-221.2kJ/mol
��C��s��+O2��g���TCO2��g����H3=-393.6kJ/mol
���H���H1����H2����H3֮��Ĺ�ϵ�ǣ���H=2��H3-��H2-��H1����Ӧ2NO��g��+2CO��g���TN2��g��+2CO2��g�����ڷ��ȷ�Ӧ��������ȡ����ȡ���
��2��Ϊ�˲ⶨ��ij�ִ��������µķ�Ӧ���ʣ�ij�о���ѧϰС���ڼ�����Ա��ָ���£��ռ�һ����������β����������ת�����Ӧ������ij�¶��£������崫������ò�ͬʱ���NO��COŨ�Ȳ������ݼ�¼���£�
| ʱ��/s | 0 | 2 | 3 | 4 |
| C��NO0����10-4mol•L-1�� | 20.0 | 3.00 | 2.00 | 2.00 |
| C��CO������10-3mol•L-1�� | 2.70 |
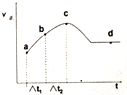
��3����С�黹�о�������ȡ����ݵ��ܱ�������ͨ��CO��NOʹ����һ�������·�����Ӧ���������Ӧ����������ͼ��ʾ��t1=��t2����
��NO��ת������a��b�κ�b��c�ε�ת���ʹ�ϵΪ����a-b����b-c�����������������=������
��a��b��c��d�ĸ��㣬�ĸ����ʾ��ѧ��Ӧ�ﵽƽ��״̬d��
�۽���c��ǰ�����ʱ仯��ԭ������һ�����ȷ�Ӧ��һ��ʼ��Ӧ�ų�����ʹ��ϵ���¶����ߣ���ʱ�¶���Ӱ�췴Ӧ���ʵ���Ҫ���أ����¶ȵ����ߣ���Ӧ���ʼӿ죬����һ��ʼʱ��Ӧ����������c����淴Ӧ�Ľ��з�Ӧ���Ũ�ȼ��٣���Ӧ��Ũ�ȵı仯��Ӱ�컯ѧ��Ӧ���ʵ���Ҫ���أ�����c���Ӧ���ʼ��٣���
| A�� | 3.4gNH3�к�N-H����ĿΪ0.2NA | |
| B�� | ��ϩ�ͻ����飨C2H4����ɵ�28g��������к���2NA����ԭ�� | |
| C�� | 15.6gNa2O2������CO2��Ӧʱ��ת�Ƶ�����Ϊ0.2NA | |
| D�� | ��0.1mol�Ȼ�������1Lˮ�У�������Һ����0.1NA��Fe3+ |
| A�� | ������ĵ���ʽ | |
| B�� | ���������е���AgNO3��Һ�������е���Ԫ�أ�Br-+Ag+�TAgBr�� | |
| C�� | ��������KOH��Һ��Ӧ��CH3CH2Br+H2O$��_{��}^{KOH}$C2H5OH+KBr | |
| D�� | ����������ȡ�Ҷ����������¼�������ȥ���ӳɡ�ˮ�� |
| A�� | CH2=CH-CH2-CH3��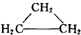 | B�� | 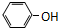 �� ��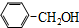 | ||
| C�� | ������ɶ�Ϊ CnH2nO2 ������ | D�� | CH3CH2Cl��CH3CH2CH2Cl |
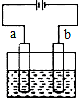
| A�� | �������Һ��Cl-��a�缫Ǩ�� | |
| B�� | ��װ���ܽ���ѧ��ת��ɵ��� | |
| C�� | b�缫�Ϸ���������Ӧ | |
| D�� | ��bΪ������b�缫�ķ�ӦʽΪ��Fe-2e-�TFe2+ |