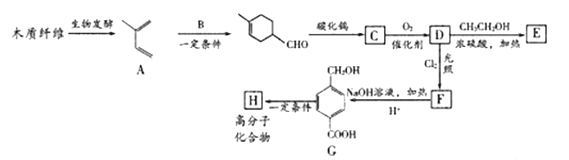
��Ŀ����
����Ŀ���Ѽ��仯�����ڻ�����ҽҩ�����ϵ��������Ź㷺��Ӧ�á�
(1)��̬��ԭ�ӵļ۵����Ų�ʽΪ_____________������ͬ���ڵ�Ԫ���У���̬ԭ�ӵ�δ�ɶԵ�����������ͬ����____________�֡�
(2)�ѱȸ��ᡢ����Ӳ����һ�����˵Ľṹ���ϣ��ѵ�Ӳ�ȱ������ԭ����_________��
(3)��Ũ��TiCl3��������Һ�м������ѣ���ͨ��HCl�����ͣ��ɵõ���λ��Ϊ6�����ΪTiCl3��6H2O����ɫ���壬�þ�����������������ʵ���֮��Ϊ1��5�����������ӵĻ�ѧʽΪ___________��
(4)����Ľṹ����M�ܴ���ϩ����ϩ������ϩ�ľۺϣ���ṹ����ͼ��ʾ��
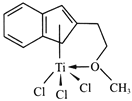
�����M��Ԫ���У��縺��������_________(������)��
��M��̼ԭ�ӵ��ӻ���ʽΪ____________��
��M���________(�����)��
a���м� b���Ҽ� c�����Ӽ� d����λ��
(5)���ʯ(TiO2)�Ǻ��ѵ���Ҫ����֮һ���侧���ṹ(��������ͬλ�õ�ԭ����ͬ)��ͼ��ʾ��
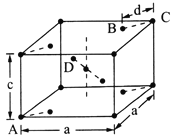
��A��B��C��D 4������������ԭ����________(�����)��
����A��B��C��ԭ������ֱ�ΪA(0��0��0)��B(0.69a��0.69a��c)��C(a��a��c)����D��ԭ������ΪD(0.19a��____��___)���������ļ���d=______(�ô���ʽ��ʾ)��
���𰸡� 3d24s2 3 Tiԭ�ӵļ۵�������Al�࣬��������ǿ [TiCl(H2O)5]2+ �� sp2 sp3 c BD 0.81a 0.5c ![]()
�����������������(1)��ԭ�Ӻ�����22�����ӣ����ݺ�������Ų�����д��̬��ԭ�ӵļ۵����Ų�ʽ����̬ԭ�ӵ�δ�ɶԵ�����Ϊ2������������δ�ɶԵ�����Ϊ2��Ԫ����Ge��Se��Ni����3����(2) Tiԭ�ӵļ۵�������4����ԭ�ӵļ۵�������3��(3).��λ��Ϊ6��������������ʵ���֮��Ϊ1��5��������������1����ԭ�ӡ�5��ˮ���ӣ�(4) �����M��Ԫ����Ti��C��H��O��Cl���ǽ�����Խǿ�縺��Խ��M����˫��̼�͵���̼ԭ���������۵���Ϊ�Ҽ���˫����1����������1��������������M�Ľṹͼ��������λ����(5) �ٸ��ݾ�̯ԭ�����й���ԭ��![]() ����������ͬλ�õ�ԭ����ͬ����������ԭ�ӱ���1:2�����������ݾ����ṹ����Dԭ�����ꣻ����ͼʾ��
����������ͬλ�õ�ԭ����ͬ����������ԭ�ӱ���1:2�����������ݾ����ṹ����Dԭ�����ꣻ����ͼʾ��![]()
������(1)��ԭ�Ӻ�����22�����ӣ���̬��ԭ�ӵļ۵����Ų�ʽΪ3d24s2����̬��ԭ�ӵ�δ�ɶԵ�����Ϊ2������������δ�ɶԵ�����Ϊ2��Ԫ�ػ���3�֣��ֱ���Ge��Se��Ni�� (2) Tiԭ�ӵļ۵�������4����ԭ�ӵļ۵�������3��Tiԭ�ӵļ۵�������Al�࣬��������ǿ�������ѵ�Ӳ�ȱ�������(3).��λ��Ϊ6��������������ʵ���֮��Ϊ1��5��������������1����ԭ�ӡ�5��ˮ���ӣ����Ը�������ӵĻ�ѧʽΪ[TiCl(H2O)5]2+��(4) �����M��Ԫ����Ti��C��H��O��Cl������O�ķǽ�������ǿ���ǽ�����Խǿ�縺��Խ�����Ե縺��������������M����˫��̼�͵���̼ԭ������������M��̼ԭ�ӵ��ӻ���ʽΪsp2�� sp3���۵���Ϊ�Ҽ���˫����1����������1��������������M�Ľṹͼ��������λ����û�����Ӽ�����ѡc��(5) �ٸ��ݾ�̯ԭ�����й���ԭ��![]() ����������ͬλ�õ�ԭ����ͬ����������ԭ�ӱ���1:2����֪��ԭ����BD�������ݾ����ṹ����A��B��C��ԭ������ֱ�ΪA(0��0��0)��B(0.69a��0.69a��c)��C(a��a��c)����Dԭ��������(0.19a��0.81a��0.5c)������ͼʾ��
����������ͬλ�õ�ԭ����ͬ����������ԭ�ӱ���1:2����֪��ԭ����BD�������ݾ����ṹ����A��B��C��ԭ������ֱ�ΪA(0��0��0)��B(0.69a��0.69a��c)��C(a��a��c)����Dԭ��������(0.19a��0.81a��0.5c)������ͼʾ��![]() ����d=
����d=![]() ��
��

 ����������ϵ�д�
����������ϵ�д�����Ŀ��ij�������ĺ���������Ҫ�����������������ʼ����ǵ���������в��ֲ�������ͼ�����ʡ�Ϊ�˴Ӻ��������л���������������µĹ�������:

������������ȫ����ʱ��Һ��pH����:
������ | Al(OH)3 | Fe(OH)3 | Fe(OH)2 | Ni(OH)2 |
pH | 5.2 | 3.2 | 9.7 | 9.2 |
�ش���������:
(1)�����Ŀ����Ϊ�˳�ȥ____________
(2)ijѧϰС����ʵ������ģ����������,����ϴ�������ٵIJ���_________
(3)����H2O2������һ��ʱ������У����µ��¶Ȳ��˹��ߵ�ԭ��Ϊ______,����H2O2������Ӧ�����ӷ���ʽΪ___________������H2O2���º��pH,���з�Χ��������______(��ѡ�����)��
A.1.8~3.2 B.2.0~3.0 C.3.3~5.2 D.9.2~9.7
(4)�����۵���Ҫ�ɷ�Ϊ_______,��Һ�۵�pHΪ2~3��Ŀ��Ϊ________
(5)Ϊ�ⶨ��Ʒ��NiSO4��7H2O���ȣ�ijͬѧ��һ��������Ʒ����ˮ,�������м���������BaCl2��Һ,���ˡ�ϴ�ӳ���������,����������ͨ�����㷢�ֲ�Ʒ��NiSO4��7H2O��������������100%,����ܵ�ԭ��Ϊ____________________