��Ŀ����
11��ԭ�������������������Ԫ��A��B��C��D�ֱ��ڵ�һ���������ڣ���Ȼ���д��ڶ���A�Ļ����Bԭ�Ӻ��������6�ֲ�ͬ���˶�״̬��B��C���γ����������η��ӣ�D�Ļ�̬ԭ�ӵ������ܲ�ֻ��һ�����ӣ������ܲ���ѳ������ӣ���ش��������⣺��1��������Ԫ���е縺������Ԫ�أ����̬ԭ�ӵļ۵����Ų�ͼΪ
 ����һ��������С��Ԫ����Cu����Ԫ�ط��ţ���
����һ��������С��Ԫ����Cu����Ԫ�ط��ţ�����2��C���������ǰ����Ԫ�طֱ���A�γɵĻ�����е��ɸߵ��͵�˳����HF��HI��HBr��HCl���ѧʽ����������˵ݱ���ɵ�ԭ����HF����֮���γ������ʹ���۷е�ϸߣ�HI��HBr��HCl����֮��ֻ�з��»�������Է�������Խ���»���Խ�е�Խ�ߣ���
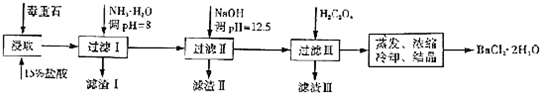
��3��BԪ�ؿ��γɶ��ֵ��ʣ�һ�־���ṹ��ͼһ��ʾ����ԭ�ӵ��ӻ�����Ϊsp2����һ�ֵľ�����ͼ����ʾ���þ����Ŀռ�������Ϊ34%�����˾����е��ⳤΪ356.6pm����˾������ܶ�Ϊ3.5g•cm-3��������λ��Ч���֣�����$\sqrt{3}=1.732$��
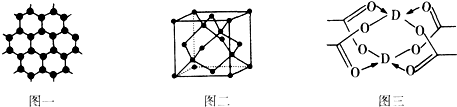
��4��DԪ���γɵĵ��ʣ��侧��Ķѻ�ģ��Ϊ�����������ܶѻ���D�Ĵ����ξ���ֲ��ṹ��ͼ�����þ����к��еĻ�ѧ���Ǣ٢ڢۣ���ѡ����ţ���
�ټ��Լ� �ڷǼ��Լ� ����λ�� �ܽ�����
��5����D����������Һ�еμӹ�����ˮ���۲쵽�������������γ���ɫ�����������μӰ�ˮ�������ܽ⣬�õ�����ɫ������Һ��
��д���������̵����ӷ���ʽ��Cu2++2NH3•H2O�TCu��OH��2��+2NH4+��Cu��OH��2+4NH3•H2O�T[Cu��NH3��4]2++2OH-+4H2O��
���� ԭ�������������������Ԫ��A��B��C��D�ֱ��ڵ�һ���������ڣ�Bԭ�Ӻ��������6�ֲ�ͬ���˶�״̬����������6�����ӣ���BΪ̼Ԫ�أ���Ȼ���д��ڶ���A�Ļ������AΪ��Ԫ�أ�D�Ļ�̬ԭ�ӵ������ܲ�ֻ��һ�����ӣ������ܲ���ѳ������ӣ�Dԭ����Χ�����Ų�Ϊ3d104s1����DΪͭԪ�أ����ԭ��������֪��Cֻ�ܴ��ڵ������ڣ�B��C���γ����������ͷ��ӣ���CΪ��Ԫ�أ�
��1������Ԫ���е縺��������Cl�����̬ԭ�ӵļ۵����Ų�Ϊ3s23p5���������ԭ�������ع����۵����Ų�ͼ��������Խǿ����һ������ԽС��
��2��HF���Ӽ����������е���ߣ�����±����������Է���������������е����ߣ�
��3��ͼһΪƽ��ṹ�������״�ṹ��̼̼������Ϊ120�㣬ÿ��̼ԭ�Ӷ������3��̼ԭ�ӣ�̼ԭ�Ӳ�ȡsp2�ӻ���
���ݾ�̯�����㾧����Cԭ����Ŀ����̼ԭ��ֱ��Ϊa�����㾧����Cԭ���������̼ԭ������Χ��4��ԭ���γ���������ṹ������̼ԭ�����������嶥��ԭ�����ڣ������ߴ�����Խ����ϣ���Ϊ�Խ��߳���$\frac{1}{4}$������Խ��߳�Ϊ4a�����ⳤΪ$\frac{4\sqrt{3}}{3}$a�������Ϊ��$\frac{4\sqrt{3}}{3}$a��3���ټ��㾧������������ռ�������=$\frac{ԭ�������}{�������}$��100%��
���ݾ�������ԭ����Ŀ�����㾧���������ٸ��ݦ�=$\frac{m}{V}$���㾧���ܶȣ�
��4������CuΪ�����������ܶѻ������ͼ������ͭ����ľֲ��ṹ��֪��̼��̼֮���γɷǼ��Լ���C����֮���γɼ��Լ���������ͭ����֮������λ����
��5������ͭ��Һ�м��백ˮ�����������ͭ��ɫ�����������μӰ�ˮ�������ܽ⣬�õ��İ���ͭ�����ӣ���ҺΪ����ɫ������Һ��
��� �⣺ԭ�������������������Ԫ��A��B��C��D�ֱ��ڵ�һ���������ڣ�Bԭ�Ӻ��������6�ֲ�ͬ���˶�״̬����������6�����ӣ���BΪ̼Ԫ�أ���Ȼ���д��ڶ���A�Ļ������AΪ��Ԫ�أ�D�Ļ�̬ԭ�ӵ������ܲ�ֻ��һ�����ӣ������ܲ���ѳ������ӣ�Dԭ����Χ�����Ų�Ϊ3d104s1����DΪͭԪ�أ����ԭ��������֪��Cֻ�ܴ��ڵ������ڣ�B��C���γ����������ͷ��ӣ���CΪ��Ԫ�أ�
��1������Ԫ���е縺��������Cl�����̬ԭ�ӵļ۵����Ų�Ϊ3s23p5�����̬ԭ�ӵļ۵����Ų�ͼΪ ������Ԫ����ֻ��CuΪ����������Ϊ�ǽ�������Cu�ĵ�һ��������С��
������Ԫ����ֻ��CuΪ����������Ϊ�ǽ�������Cu�ĵ�һ��������С��
�ʴ�Ϊ�� ��Cu��
��Cu��
��2��HF����֮���γ������ʹ���۷е�ϸߣ�HI��HBr��HCl����֮��ֻ�з��»�������Է�������Խ���»���Խ�е�Խ�ߣ����е��ɸߵ��͵�˳����HF��HI��HBr��HCl��
�ʴ�Ϊ��HF��HI��HBr��HCl��HF����֮���γ������ʹ���۷е�ϸߣ�HI��HBr��HCl����֮��ֻ�з��»�������Է�������Խ���»���Խ�е�Խ�ߣ�
��3��ͼһΪƽ��ṹ�������״�ṹ��̼̼������Ϊ120�㣬ÿ��̼ԭ�Ӷ������3��̼ԭ�ӣ�̼ԭ�Ӳ�ȡsp2�ӻ���
һ�������к�̼ԭ����Ϊ8��$\frac{1}{8}$+6��$\frac{1}{2}$+4=8����̼ԭ��ֱ��Ϊa��������Cԭ�������=8��$\frac{4}{3}$�У�$\frac{a}{2}$��3��̼ԭ������Χ��4��ԭ���γ���������ṹ������̼ԭ�����������嶥��ԭ�����ڣ������ߴ�����Խ����ϣ���Ϊ�Խ��߳���$\frac{1}{4}$������Խ��߳�Ϊ4a�����ⳤΪ$\frac{4\sqrt{3}}{3}$a�������Ϊ��$\frac{4\sqrt{3}}{3}$a��3�������ռ�������={[8��$\frac{4}{3}$�У�$\frac{a}{2}$��3]�£�$\frac{4\sqrt{3}}{3}$a��3}��100%��34%��
��������Ϊ$\frac{8��12}{6.02��1{0}^{23}}$g�����˾����е��ⳤΪ356.6pm�������Ϊ��356.6��10-10cm��3����˾������ܶ�Ϊ $\frac{8��12}{6.02��1{0}^{23}}$g�£�356.6��10-10cm��3=3.5g��cm-3��
�ʴ�Ϊ��sp2��34%��3.5��
��4������CuΪ�����������ܶѻ������ͼ������ͭ����ľֲ��ṹ��֪��̼��̼֮���γɷǼ��Լ���C����֮���γɼ��Լ���������ͭ����֮������λ�����侧���к��м��Լ����Ǽ��Լ�����λ����
�ʴ�Ϊ�������������ܶѻ����٢ڢۣ�
��5������ͭ��Һ�м��백ˮ�������ɫ�����������μӰ�ˮ�������ܽ⣬�õ�����ɫ������Һ���йط�Ӧ�����ӷ���ʽΪCu2++2NH3•H2O�TCu��OH��2��+2NH4+��Cu��OH��2+4NH3•H2O�T[Cu��NH3��4]2++2OH-+4H2O��
�ʴ�Ϊ�������γ���ɫ�����������μӰ�ˮ�������ܽ⣬�õ�����ɫ������Һ��Cu2++2NH3•H2O�TCu��OH��2��+2NH4+��Cu��OH��2+4NH3•H2O�T[Cu��NH3��4]2++2OH-+4H2O��
���� �����Ƕ����ʽṹ�Ŀ��飬��Ŀ�ۺ��Խϴ��漰Ԫ���ƶϡ���������Ų�������ṹ�뻯ѧ�����ӻ������������������ȣ��Ƕ�ѧ���ۺ������Ŀ��飬��3���м���Ϊ�״��㡢�Ѷȣ���Ҫѧ���߱�һ���Ŀռ���������ѧ�����������Ѷ��еȣ�

 �߽�������ϵ�д�
�߽�������ϵ�д������ڿ�����ȼ�� �ں���Ļ������ڿ��������� ���������ƺ����ᷴӦ �������ڿ�������ȫȼ�� ��ͭ����Ũ���ᷴӦ��
| A�� | ֻ�Т٢ۢ� | B�� | ֻ�Т٢� | C�� | ֻ�Т٢ۢܢ� | D�� | �٢ڢۢܢ� |
��1������ʯ�������ȡǰ������ĥ��Ŀ��������Ӵ�����Ӷ�ʹ��Ӧ���ʼӿ죮ʵ������37%����������15%�����ᣬ����Ͳ���ʹ�����������е�ac��
a���ձ� b������ƿ c�������� d���ζ���
��2��
| Ca2+ | Mg2+ | Fe3+ | |
| ��ʼ����ʱ��pH | 11.9 | 9.1 | 1.9 |
| ��ȫ����ʱ��pH | 13.9 | 11.1 | 3.2 |
��֪��Ksp��BaC2O4��=1.6��10-7��Ksp��CaC2O4��=2.3��10-9
��3�����ü�����ζ����ɲⶨBa2+�ĺ�����ʵ����������У�
��֪��2CrO42-+2H+�TCr2O72-+H2O Ba2++CrO42-�TBaCrO4��
�������ȡxmlһ��Ũ�ȵ�Na2CrO4��Һ����ƿ�У��������ָʾ������b mol•L-1�����Һ�ζ����յ㣬��õμ��������ΪV0mL��
�������ȡy mLBaCl2��Һ����ƿ�У�����x mL�벽�����ͬŨ�ȵ�Na2CrO4��Һ����Ba2+��ȫ�������ټ������ָʾ������b mol•L-1�����Һ�ζ����յ㣬��õμ���������ΪV1mL����BaCl2��Һ��Ũ��Ϊ$\frac{��{V}_{0}-{V}_{1}��b}{y}$mol•L-1��
| A�� | ��101kPaʱ��1mol������ȫȼ��ʱ���ų������������������ʵ�ȼ���� | |
| B�� | �����£���ȼ��ȼ�շų������� | |
| C�� | ��25�桢1.01��105Paʱ��1mol��������ȫȼ�������ȶ���������ʱ���ų������� | |
| D�� | ȼ�����滯ѧ����ʽǰ�Ļ�ѧ�������ĸı���ı� |
��Al$\frac{ϡ����}{\;}$AlCl3$\stackrel{NaOH��Һ}{��}$Al��OH��3��
��Al$\stackrel{NaOH��Һ}{��}$Na[Al��OH��4]$\frac{ϡ����}{\;}$Al��OH��3
��
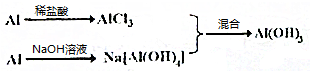
| ���� | ����1mol Al��OH��3�����Լ������ʵ��� | |
| n HCl/mol | n NaOH/mol | |
| �� | ||
| �� | ||
| �� | ||
| A�� | �ֻ����� | B�� | ��ѧ���� | C�� | ʯӢ���� | D�� | ��ɫ���� |
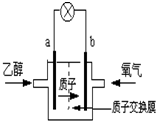 2004������ʥ·��˹��ѧ������һ�����͵��Ҵ���أ����û� ���������ܼ�����200��C����ʱ���磬�Ҵ���رȼ״����Ч�ʸ߳�32���Ҹ���ȫ������ܷ�ӦΪ��C2H5OH+3O2�T2CO2+3H2O�����ʾ����ͼ������˵������ȷ���ǣ�������
2004������ʥ·��˹��ѧ������һ�����͵��Ҵ���أ����û� ���������ܼ�����200��C����ʱ���磬�Ҵ���رȼ״����Ч�ʸ߳�32���Ҹ���ȫ������ܷ�ӦΪ��C2H5OH+3O2�T2CO2+3H2O�����ʾ����ͼ������˵������ȷ���ǣ�������| A�� | a��Ϊ��صĸ��� | |
| B�� | ��ع���ʱ������b���ص��߾������ٵ�a�� | |
| C�� | ��ع���ʱ��1mol�Ҵ�����ԭʱ����6mol����ת�� | |
| D�� | ��������ĵ缫��ӦΪ��4H++O2+4e-��2H2O |