��Ŀ����
��10�֣����Ṥҵ�����е�β����NO2��NO�Ļ�����壩ͨ�����ô�����Һ���գ�
2NO2��Na2CO3��NaNO3��NaNO2��CO2�� ��
NO��NO2��Na2CO3��2NaNO2��CO2�� ��
��1�������Ṥҵβ�����չ����У��ڱ�״���£�ÿ����44.8L������̼���壬��������β�� L��
��2������ҵ����21.2%�Ĵ�����Һ�������������е�β��ʱ����Ҫ����10 kg��Ũ�ȵĴ�����Һ����Ҫ���Na2CO3��10H2O�������Ƕ��٣�
��3��Ҫ��֤���Ṥҵ��β��������գ�n(NO2)��n(NO)Ӧ������ֹ�ϵ��
��4������10 kg 21.2%�Ĵ�����Һ����������NO2��NO���� ��״����ÿ����448L������̼ʱ��������Һ����880 g��
��״����ÿ����448L������̼ʱ��������Һ����880 g��
��������NaNO3��NaNO2���������Ƕ��٣�
��20��ʱ��10 kg 21.2%�Ĵ�����Һ������ȫ��������6.88 kgˮ����ȴ��5�棬������NaNO2���������Ƕ��٣�����֪5��ʱ��NaNO2 ���ܽ��Ϊ71.2g/100gˮ������NaNO3��Ӱ�죩��
2NO2��Na2CO3��NaNO3��NaNO2��CO2�� ��
NO��NO2��Na2CO3��2NaNO2��CO2�� ��
��1�������Ṥҵβ�����չ����У��ڱ�״���£�ÿ����44.8L������̼���壬��������β�� L��
��2������ҵ����21.2%�Ĵ�����Һ�������������е�β��ʱ����Ҫ����10 kg��Ũ�ȵĴ�����Һ����Ҫ���Na2CO3��10H2O�������Ƕ��٣�
��3��Ҫ��֤���Ṥҵ��β��������գ�n(NO2)��n(NO)Ӧ������ֹ�ϵ��
��4������10 kg 21.2%�Ĵ�����Һ����������NO2��NO����
 ��״����ÿ����448L������̼ʱ��������Һ����880 g��
��״����ÿ����448L������̼ʱ��������Һ����880 g����������NaNO3��NaNO2���������Ƕ��٣�
��20��ʱ��10 kg 21.2%�Ĵ�����Һ������ȫ��������6.88 kgˮ����ȴ��5�棬������NaNO2���������Ƕ��٣�����֪5��ʱ��NaNO2 ���ܽ��Ϊ71.2g/100gˮ������NaNO3��Ӱ�죩��
��1��89.6L��2�֣� ��2��5.72kg��2�֣�
��3��n(NO2)��n(NO)��
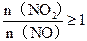 ��2�֣�
��2�֣���4����17��2
 3��2�֣�
3��2�֣���1.013kg��2�֣�
��������ȷ��ͬ�����֣�
��

��ϰ��ϵ�д�
�����Ŀ
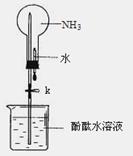
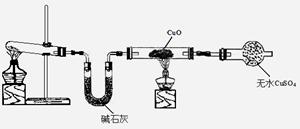
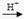 Cu + Cu2+����
Cu + Cu2+����
 2O5��5KCl��������������������
2O5��5KCl��������������������