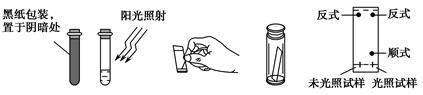
��Ŀ����
�����к�̼�����Ԫ�ء���ѧ��ȤС���ij������Ʒ������̽��������Ҫ��ش��������⡣
��.������̼����Ԫ�صĶ��Լ���
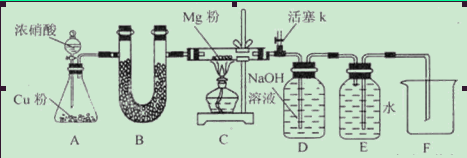
����ͼװ�ý���ʵ��(�г���������ȥ,��ӿ���ĸ����)����ʵ��̼����Ԫ�صļ��顣
(1)����X��������������;װ�âۢ����Լ���ͬ,װ�â���ʢ�ŵ��Լ����� ������
(2)д�����з�Ӧ�Ļ�ѧ����ʽ����������������������������������������
(3)�������װ�âۢܢ�,�ܷ�ȷ��������Ʒ��̼Ԫ�صĴ���?��������,������������������������������������������������
��.������̼�������������IJⶨ
(4)��ͬѧ��Ϊ,����װ�ÿ��Դ��Բⶨ��Ʒ��̼�ĺ�������ȡ��Ʒw1 g����ʵ��,��ַ�Ӧ��,���װ�â������ɵij���Ϊw2 g,����Ʒ��̼����������Ϊ������(�ú�w1��w2��ʽ�ӱ�ʾ)��
(5)��ͬѧ��Ϊ,��һ������Ʒ��ַ�Ӧ��,��װ�â��м�������Ȼ�����Һ,���ݳ����������Լ�����Ʒ�������������,�˷�����õĽ��������(�ƫ��ƫС��);��Ҫ�����Ԫ�غ����IJⶨ����,�ڲ��ı�ʵ��ԭ����ǰ����,���Բ�ȡ��һ�ִ�ʩ������������������������������
(1)��Һ©�������Ը��������Һ(����ˮ)
(2)2H2O2 2H2O+O2��
2H2O+O2��
(3)���ܡ�SO2Ҳ��ʹ����������Һ�����
(4)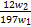
(5)ƫС����Ӧ������,����ͨ������һ��ʱ��;��������Һ���ȵ���Ϊ��ɫ
����

 ������ȫ��������ϵ�д�
������ȫ��������ϵ�д�ijͭ��ʯ��ͭԪ�غ����ϵͣ��Һ�������þ���Ƶ����ʡ�ijС����ʵ�������ý�������ȡ���Ʊ�����ͭ��
(1)������Ϊ________���������õ��IJ����������ձ���________��
(2)�������������ҪĿ����________������ͭԪ�ء�
(3)С���Ա����CuSO4��Һ��Na2CO3��Һ��Ϸ�Ӧ���Ʊ�������ľ�ķ�����Cu2(OH)2CO3����Һ�����ʵ�鷢��������ɫ����Һ��ɫ���в��죬�������ϱ��������������������Ʋ�ͬʹ���л��н϶�Cu(OH)2��Cu4(OH)6SO4��
��֪Cu(OH)2��Cu2(OH)2CO3��Cu4(OH)6SO4��������ˮ����������ֽ��¶�����Ϊ80 �桢200 �桢300 �档
���ʵ���������Һ�ɷ֣���ɱ������ݡ�
��ѡ�Լ���2 mol��L��1���ᡢ1 mol��L��1 H2SO4��0.1 mol��L��1 NaOH��Һ��0.1 mol��L��1 BaCl2��Һ������ˮ����������Ʒ��ѡ��
| ʵ�鲽�� | Ԥ������ͽ��� |
| ����1��ȡ��������Һ�����ˣ����ϴ�Ӻ�ȡ�������Թ��У�______________________ | ____________��˵������Һ�л���Cu4(OH)6SO4 |
| ����2����ȡ��������Һ���Թ��У�____________________ | ____________��˵������Һ�л���Cu(OH)2 |
(4)����ʵ����Ҫ100 mL 0.5 mol��L��1��CuSO4��Һ������ʱ���ȡ________g CuSO4��5H2O(��Է���������250)��
��ͼ1��ʾ��ʵ��������ȡ�����һ�ּ���װ�á�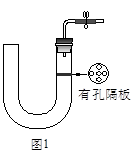
��1��������������������Եķ���_______________________��
��2��������ͼ1��ʾװ�ÿ�����ȡ���Ӧ��״����������Ӧ�Ƿ���Ҫ��������________________________���塣
ijͬѧ�����ͼ2��ʾװ�ã��ô�������16.9%ϡ���ᷴӦ��ȡNO���岢̽�����������ļ�̬����ش��й����⡣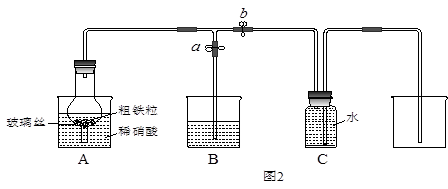
��3����֪16.9%ϡ������ܶ�Ϊ1.10g/cm3���������ʵ���Ũ��Ϊ____________��������������λС������������63%����������16.9%ϡ����500mL������IJ��������в��������ձ��� ��
��4������ֹˮ��a���ر�ֹˮ��bʱ��Aװ�õĸ�����й۲쵽��������_______________________��Bװ���ձ���Һ���������__________________________________����Aװ�������弸����ɫʱ����ֹˮ��b���ر�ֹˮ��a������Cװ���ռ�NO���塣
��5����֪���з�Ӧ���Է�����Fe2O3+3KNO3+4KOH 2K2FeO4+3KNO2+2H2O����Aװ���е�ϡ���ἴʹ����Ũ���ᣬҲ��������+6�۵����Ļ������ԭ����________��
2K2FeO4+3KNO2+2H2O����Aװ���е�ϡ���ἴʹ����Ũ���ᣬҲ��������+6�۵����Ļ������ԭ����________��
a��HNO3�������Ա�KNO3��
b����Ӧ���¶Ȳ���
c��HNO3�����ȶ��Ա�KNO3��
d��FeO42�����ܴ�����������Һ��
��6������������ҩƷ���Թܺͽ�ͷ�ιܣ�0.1mol/LKSCN��Һ��0.2mol/L����KMnO4��Һ��0.1mol/LKI��Һ����ˮ�ȡ��������һ����ʵ�飬̽��Aװ���ձ�����ȫ��Ӧ�������ܵļ�̬����д����ʵ�鱨�棺
| ʵ�鲽�� | ���� | ��������� |
| ��һ�� | ȡ����Һ��װ���Թܣ����� ���е��뼸��KSCN��Һ�� | |
| �ڶ��� | | ����Һ��ɫ��ȥ����˵������Fe2+�� �������Ա仯����˵������Fe2+�� |
��ͼ��ʵ�����Ʊ�1��2���������鲢����һϵ�����ʵ���װ�ã����ȼ��г��豸���ԣ���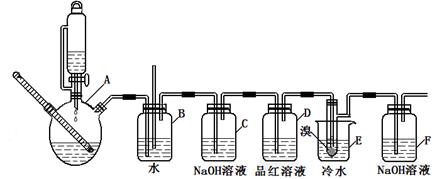
�й������б����£�
| | �Ҵ� | 1,2-�������� | ���� |
| ״̬ | ��ɫҺ�� | ��ɫҺ�� | ��ɫҺ�� |
| �ܶȣ�g/cm3 | 0.79 | 2.2 | 0.71 |
| �е㣯�� | 78.5 | 132 | 34.6 |
| �۵㣯�� | һl30 | 9 | -1l6 |
��1��A��ҩƷΪ1:3����ˮ�Ҵ���Ũ������Һ��д���Ʊ���ϩ�Ļ�ѧ��Ӧ����ʽ��____________________________________________________________��
��2�����巢��װ��ʹ����ͨ��Һ©����ԭ��_________________________________________��
��3��װ��D��Ʒ����Һ��������_______________��ͬʱBװ���ǰ�ȫƿ�����ʵ�����ʱE���Ƿ�����������д������ʱ������_______________________________________��
��4����Ӧ������Ӧ����ˮ��ȴװ��E������ҪĿ����___________________________�����ֲ��ܹ�����ȴ(���ñ�ˮ)����ԭ����_____________________________________________��
��5���жϸ��Ʊ���Ӧ�Ѿ������ķ�����__________________�����ѧ�����ַ�Ӧ����ʱ����ˮ�Ҵ��������������ֵ����ԭ����_______________________________________��
��6����ѧ�������װ��F�пɸ������������Ȼ�̼Һ�����ն�������壬�жϸ������Ȼ�̼Һ���Ƿ����______����ǡ�������ԭ����____________________________��