��Ŀ����
����Ŀ������������ԭ��Ӧ��2Ag��(aq)��Zn(s)===Zn2��(aq)��2Ag(s)��Ƶ�ԭ�����ͼ��ʾ����ش��������⣺

(1)�缫X�IJ�����______���������ҺY��________��
(2)���缫Ϊ��ص�________���������ĵ缫��ӦΪ____________________��X�缫�Ϸ����ĵ缫��ӦΪ_________________________________________��
(3)���·�еĵ����Ǵ�______�缫����______�缫��
(4)���õ�������缫��������Ϊ100g������һ��ʱ���пƬ����������13g���Լ��㣬���缫��������_________g��
���𰸡�Zn AgNO3 ���� Ag++e-=Ag Zn-2e-=Zn2+ X(п) �� 43.2g
��������
���ݷ�Ӧ��2Ag+(aq)+Zn(s)�TZn2+(aq)+2Ag(s)���������ڷ�Ӧ�У�Zn��������ʧ���ӣ�ӦΪԭ��صĸ������缫��ӦΪZn-2e-=Zn2+��������Ϊ�����Խ�Zn����Ag��Ag+�������ϵõ��ӱ���ԭ���缫��ӦΪAg++e=Ag���������ҺΪAgNO3 ��ԭ����У����ӴӸ��������·�������������ݵ�ʧ�����غ������������������
(1)�ɷ�Ӧ��2Ag+(aq)+Zn(s)�TZn2+(aq)+2Ag(s)����֪���ڷ�Ӧ�У�Zn��������ʧ���ӣ�ӦΪԭ��صĸ������缫��ӦΪZn-2e-=Zn2+��������Ϊ�����Խ�Zn����Ag��Ag+�������ϵõ��ӱ���ԭ���缫��ӦΪAg++e=Ag���������ҺΪAgNO3
(2)����Ϊ�����Խ�Zn����Ag��Ag+�������ϵõ��ӱ���ԭ���缫��ӦΪAg++e=Ag��X�缫ΪZnΪԭ��صĸ�����пʧ���ӷ���������Ӧ���缫��ӦZn-2e-�TZn2+��
(3)ԭ����У����ӴӸ��������·������������������Zn�������·����Ag����
(4)��ԭ����У�������пʧ���ӱ��п���ӽ�����Һ�������������٣�����п������=60g-47g=13g�����ʵ���=![]() =0.2mol������ת�Ƶ������֪��2Ag++2e-=2Ag��Zn-2e-�TZn2+��
=0.2mol������ת�Ƶ������֪��2Ag++2e-=2Ag��Zn-2e-�TZn2+��
2Ag����Zn
21
n0.2mol
n=0.4mol��������������=0.4mol��108g/mol=43.2g�������缫���ص�����Ϊ43.2g��

 ���ʿ��ÿ��ֳɳ�ϵ�д�
���ʿ��ÿ��ֳɳ�ϵ�д�����Ŀ������N2O5Ϊ��������һ�����͵���ɫ�����������ں��ܲ��ϡ�ҽҩ�ȹ�ҵ�еõ��㷺Ӧ�á��ش���������
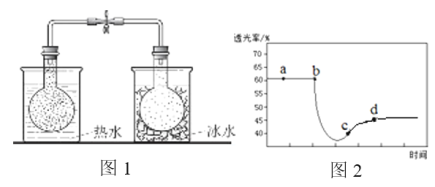
��1��1840�� Devil�ø��������ͨ����������������õ�N2O5���÷�Ӧ������������һ�����壬�����ʽΪ___________��
��2��F. Daniels�������ò�ѹ���ڸ��Է�Ӧ�����о���25��ʱN2O5(g)�ֽⷴӦ��

����NO2����ΪN2O4�ķ�Ӧ����Ѹ�ٴﵽƽ�⡣��ϵ����ѹǿp��ʱ��t�ı仯���±���ʾ��t=��ʱ��N2O5(g)��ȫ�ֽ⣩��
t/min | 0 | 40 | 80 | 160 | 260 | 1300 | 1700 | �� |
p/kPa | 35.8 | 40.3 | 42.5. | 45.9 | 49.2 | 61.2 | 62.3 | 63.1 |
����֪��2N2O5(g)��2N2O4(g)+O2(g) ��H1=4.4 kJ��mol1 ��2NO2(g)��N2O4(g) ��H 2=55.3 kJ��mol1����ӦN2O5(g)��2NO2(g)+1/2O2(g)�Ħ�H =_______ kJ��mol1��
���о�������N2O5(g)�ֽ�ķ�Ӧ����v=2��10-3��P(N2O5)(KPa/min)��t=62 minʱ�������ϵ��P(O2)=2.9 kPa�����ʱ��P(N2O5)=________ kPa��v=_______kPa��min1��
������߷�Ӧ�¶���35�棬��N2O5(g)��ȫ�ֽ����ϵѹǿp��(35��)____63.1 kPa������ڡ������ڡ���С�ڡ�����
��25��ʱN2O4(g)![]() 2NO2(g)��Ӧ��ƽ�ⳣ��Kp=_______kPa��KpΪ�Է�ѹ��ʾ��ƽ�ⳣ��������������1λС������
2NO2(g)��Ӧ��ƽ�ⳣ��Kp=_______kPa��KpΪ�Է�ѹ��ʾ��ƽ�ⳣ��������������1λС������
��3�����ڷ�Ӧ2N2O5(g)��4NO2(g)+O2(g)��R.A.Ogg������·�Ӧ���̣�
��һ�� N2O5![]() NO2+NO3 ����ƽ��
NO2+NO3 ����ƽ��
�ڶ��� NO2+NO3��NO+NO2+O2 ����Ӧ
������ NO+NO3��2NO2 �췴Ӧ
���пɽ�����Ϊ�ڶ�����Ӧ��Ӱ���һ����ƽ�⡣���б�����ȷ����_______������)��
A��v(��һ�����淴Ӧ)��v(�ڶ�����Ӧ) B����Ӧ���м����ֻ��NO3
C���ڶ�����NO2��NO3����ײ��������Ч D����������Ӧ��ܽϸ�