��Ŀ����
17��ԭ��������������Ķ�����Ԫ��a��b��c��d��e�У�a������������Ϊ���������Ķ�����b��d��A2B���⻯���ΪV�η��ӣ�c��+1�����ӱ�e��-1��������8�����ӣ��ش��������⣺
��1��Ԫ��a�ĵ����Ų�ͼΪ
 ��Ԫ��e�ĵ����Ų�ʽΪ1s22s22p63s23p5��c�����ӽṹʾ��ͼΪ
��Ԫ��e�ĵ����Ų�ʽΪ1s22s22p63s23p5��c�����ӽṹʾ��ͼΪ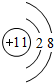
��2������ЩԪ�����γɵ�˫ԭ�ӻ�������ӵ�����Ϊһ����̼��
��3������ЩԪ���γɵ���ԭ�ӷ����У����ӵĿռ�ṹ����ֱ���ε���CO2��CS2
��4����ЩԪ���γɵ�AB�ͻ������У��侧�������������Ӿ������NaCl�����ڷ��Ӿ������CO��ÿ������һ�ֻ�ѧʽ�������õ���ʽ��ʾ�����Ӿ�����γɹ���
 ��
����5��Ԫ��c��e�γɵ�һ�ֻ������ˮ��Һ�����ڹ�ҵ����e�õ��ʣ��÷�Ӧ�Ļ�ѧ����ʽΪ2NaCl+2H2O$\frac{\underline{\;���\;}}{\;}$2NaOH+H2��+Cl2����
���� ԭ��������������Ķ�����Ԫ��a��b��c��d��e�У�a������������Ϊ���������Ķ�������a��2�����Ӳ㣬����������Ϊ4����aΪ̼Ԫ�أ�b��d��A2B���⻯���ΪV�η��ӣ�bΪ��Ԫ�أ�dΪ��Ԫ�أ�c��+1�����ӱ�e��-1��������8�����ӣ�cΪ��Ԫ�أ�eΪClԪ�أ�
��� �⣺ԭ��������������Ķ�����Ԫ��a��b��c��d��e�У�a������������Ϊ���������Ķ�������a��2�����Ӳ㣬����������Ϊ4����aΪ̼Ԫ�أ�b��d��A2B���⻯���ΪV�η��ӣ�bΪ��Ԫ�أ�dΪ��Ԫ�أ�c��+1�����ӱ�e��-1��������8�����ӣ�cΪ��Ԫ�أ�eΪClԪ�أ�
��1��Ԫ��aΪ̼Ԫ�أ������Ų�ͼΪ ��Ԫ��eΪCl�������Ų�ʽΪ 1s22s22p63s23p5��c������ΪNa+�����ӽṹʾ��ͼΪ
��Ԫ��eΪCl�������Ų�ʽΪ 1s22s22p63s23p5��c������ΪNa+�����ӽṹʾ��ͼΪ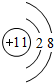 ��
��
�ʴ�Ϊ�� ��1s22s22p63s23p5��
��1s22s22p63s23p5��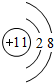 ��
��
��2������ЩԪ�����γɵ�˫ԭ�ӻ���������У�һ����̼��
�ʴ�Ϊ��һ����̼��
��3��������Ԫ����ɵ���ԭ�ӷ����У����ӵĿռ�ṹ����ֱ���ε���CO2��CS2��
�ʴ�Ϊ��CO2��CS2��
��4����ЩԪ���γɵ�AB�ͻ������У��侧�������������Ӿ������NaCl�����ڷ��Ӿ������CO���õ���ʽ��ʾNaCl���Ӿ�����γɹ��� ��
��
�ʴ�Ϊ��NaCl��CO�� ��
��
��5��Ԫ��c��e�γɵĻ�����ΪNaCl��NaCl��ˮ��Һ�����ڹ�ҵ�����������÷�Ӧ�Ļ�ѧ����ʽΪ��2NaCl+2H2O$\frac{\underline{\;���\;}}{\;}$2NaOH+H2��+Cl2����
�ʴ�Ϊ��2NaCl+2H2O$\frac{\underline{\;���\;}}{\;}$2NaOH+H2��+Cl2����
���� ���⿼��ṹ����λ�ù�ϵӦ�ã��ƶ�Ԫ���ǽ���ؼ������ػ�ѧ���������ṹ�Ŀ��飬�Ѷ��еȣ���Ҫѧ���������ջ���֪ʶ��

| ���� | ������������ | ������g�� | ���ʵ�����mol�� | Ħ��������g/mol�� |
| ���� | 0.3 | |||
| S | 1.204��1023 | |||
| CO2 | 22 | |||
| C12H22O11 | 0.25 |
��1����ҵ����������У�SO2��������ԭ��Ϊ��
2SO2��g��+O2��g��$?_{��}^{����}$2SO3��g��
T��ʱ����ij�ܱ������г���һ��SO2��g����O2��g��������������Ӧ�����SO2��g����ƽ��ת���ʣ�a������ϵ��ѹǿ��p���Ĺ�ϵ��ͼ1��ʾ��
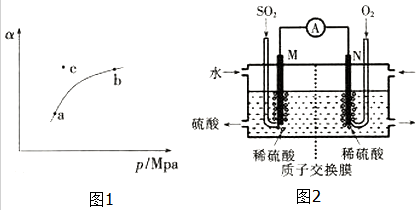
��a��b�����Ӧ��ƽ�ⳣ��K��a��=K��b�������������������=������ͬ����SO2Ũ��c��a����c��b����
��c��ʱ����Ӧ����v��������v���棩��
��2���绯ѧ������SO2��
���Ṥҵβ���е�SO2������������Ʊ����ᣬͬʱ��õ��ܣ�װ����ͼ2��ʾ���缫��Ϊ���Բ��ϣ���
��M�������ĵ缫��ӦʽΪSO2-2e-+2H2O=4H++SO42-��
����ʹ��װ�õĵ���ǿ�ȴﵽ2.0A��������ÿ����Ӧ��ͨ���״������������Ϊ0.014L����֪��1��e-��������Ϊ1.6��10-19C����
��3����Һ������SO2��
��֪������H2SO3��H2CO3�ĵ��볣�������ʾ��
| ���볣�� �� | K1 | K2 |
| H2SO3 | 1.3��10-2 | 6.3��10-8 |
| H2CO3 | 4.2��10-7 | 5.6��10-11 |
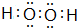 �����õ���ʽ��ʾԭ�Ӹ�����Ϊ2��1�ĸû�������γɹ���
�����õ���ʽ��ʾԭ�Ӹ�����Ϊ2��1�ĸû�������γɹ��� ��
��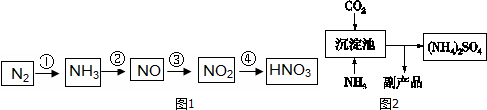
 ij��ȤС��ͬѧ�о�������Ԫ�ؼ���ijЩ������IJ������ʣ������������£�
ij��ȤС��ͬѧ�о�������Ԫ�ؼ���ijЩ������IJ������ʣ������������£�