��Ŀ����
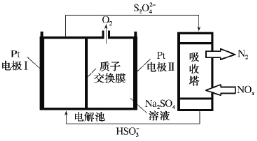
����Ŀ�������к�����������̿��������һ�֣��о����������Ի�����ӣ����ɻ��������̵������仯ģ���������ͼ��ʾ��������Կ�������SO2������˵����ȷ���ǣ� ��
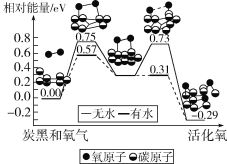
A.ÿ�һ������������0.29eV����
B.ˮ��ʹ�����ӻ��Ӧ�Ļ�ܽ���0.18eV
C.�����ӵĻ��O��O���Ķ�����C��O�������ɹ���
D.̿�ڿ����Ǵ�����SO2ת��ΪSO3��������
���𰸡�BC
��������
A����ͼ�п��Կ�������Ӧǰ1�������Ӽ�̿�ڵ�������Ϊ0.00����Ӧ�յ�ʱ�������������Ϊ-0.29eV������ÿ�һ���������ܹ��ͷ�0.29eV������A����ȷ��
B���Ա���ͬ����ˮ����ˮʱ�Ļ�ܣ���ˮʱ���Ϊ0.75eV����ˮʱ���Ϊ0.57eV������ˮ��ʹ�����ӻ��Ӧ�Ļ�ܽ���0.75eV-0.57eV = 0.18eV��B��ȷ��
C���ڷ�Ӧ�����У�����O-O���������������γ�C-O����ų����������������ӵĻ��O-O���Ķ�����C-O�������ɹ��̣�C��ȷ��
D��̿�ڿ����ɻ�����ӣ�����Ǵ�����SO2ת��ΪSO3����������̿���൱�ڴ�����D����ȷ��
��ѡBC��

����Ŀ�����и�����������Һ�п��Դ������棬�Ҽ����ͨ���Լ�X������Ӧ�����ӷ���ʽ��ȷ����
ѡ�� | ������ | �Լ�X | ������Ӧ�����ӷ���ʽ |
A | K+��Na+�� | ����HCl | H2O+H++ |
B |
| ����H2S | Fe2++H2S=FeS��+2H+ |
C | ClO-��Na+��Ca2+�� | ����CO2 | CO2+Ca2++H2O+2ClO-=CaCO3��+2HClO |
D | I-��Cl-��Na+�� | ����ϡ���� | 6I-+2 |
A.AB.BC.CD.D