��Ŀ����
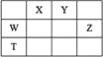
����Ŀ��ij��ɫ��Һ�п��ܺ���Na+��Ba2+��Cu2+��CO32����Cl����Mg2+�е�һ�ֻ������ӡ�
�������Һ�еμ�����ϡ��������������
��ȡ����������Һ������������Na2SO4��Һ���а�ɫ����������
��ȡ�����ϲ���Һ������������NaOH��Һ���а�ɫ����������
��1��ԭ��Һ��һ�����е�������________��һ�������е�������________������ȷ���Ƿ��е�������________��
��2�����з�Ӧ�����ӷ���ʽΪ___________________��
��3������ȡ10mL������Һ����ƿ�У�Ȼ�������Һ����μ���NaOH��Һ(��ͼ����ʾ)���μӹ����в������������������NaOH��Һ������Ĺ�ϵ��ͼ����ʾ��
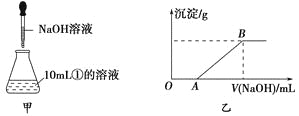
��OA�η�Ӧ�����ӷ���ʽΪ_____________________��
��AB�η�Ӧ�����ӷ���ʽΪ_______________________��
���𰸡�Ba2+��Cl����Mg2+ CO32����Cu2+ Na+ Ba2+��SO42��=BaSO4�� H����OH��=H2O Mg2����2OH��=Mg(OH)2��
��������
��ɫ��Һ��һ������Cu2+������Һ�еμ�����ϡ��������������һ��û��CO32����ȡ����������Һ������������Na2SO4��Һ���а�ɫ����������һ����Ba2+��ȡ�����ϲ���Һ������������NaOH��Һ���а�ɫ����������һ����Mg2+�����ݵ���غ㣬һ����Cl����
��1��ԭ��Һ��һ�����е�������Ba2+��Cl����Mg2+��һ�������е�������CO32����Cu2+������ȷ���Ƿ��е�������Na+��
��2�����з�Ӧ�����ӷ���ʽΪBa2+��SO42��=BaSO4����
��3��������Һ�к���Ba2+��Cl����Mg2+��H+���μ��������ƣ��������Ⱥ����������ӷ�Ӧ��Ȼ����Mg2+�����������ӷ�Ӧ������OA��ΪH����OH��=H2O��AB�η�Ӧ�����ӷ���ʽΪMg2����2OH��=Mg(OH)2����

����Ŀ�������ü�������NO2��Ⱦ�����о���CH4+2NO2 ![]() N2+CO2+2H2O����1L�ܱ������У����Ʋ�ͬ�¶ȣ��ֱ����0.50molCH4��1.2molNO2�����n(CH4)��ʱ��仯���й�ʵ�����ݼ��±���
N2+CO2+2H2O����1L�ܱ������У����Ʋ�ͬ�¶ȣ��ֱ����0.50molCH4��1.2molNO2�����n(CH4)��ʱ��仯���й�ʵ�����ݼ��±���
��� | �¶� | ʱ��/min n/mol | 0 | 10 | 20 | 40 | 50 |
�� | T1 | n(CH4) | 0.50 | 0.35 | 0.25 | 0.10 | 0.10 |
�� | T2 | n(CH4) | 0.50 | 0.30 | 0.18 | �� | 0.15 |
����˵����ȷ����
A. �����У�0~20min�ڣ�NO2�Ľ�������Ϊ0.0125 mol��L-1��min-1
B. ��ʵ�����ݿ�֪ʵ����Ƶ��¶�T12
C. 40minʱ��������T2Ӧ�������Ϊ0.18
D. 0��10min�ڣ�CH4�Ľ������ʢ�>��