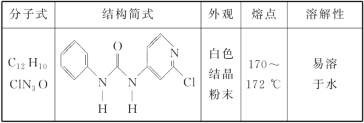
��Ŀ����
����Ŀ��������,��0.100mol��L-1NaOH��Һ�ֱ�ζ�20.00ml 0.100mol��L-1������ʹ���,�ζ���������ͼ��ʾ������˵����ȷ����( )
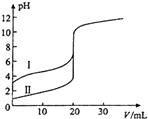
A. ���ʾ���ǵζ����������
B. pH=7ʱ,�ζ��������ĵ�V(NaOH)С�ڵζ��������ĵ�V(NaOH)
C. V(NaOH)=10.00mLʱ,������Һ��C(CH3COO-)+2C(H+)=C(CH3COOH)+2C(OH-)
D. V(NaOH)=20.00mLʱ,������Һ��C(Cl-)=C(CH3COO-)
���𰸡�B
��������
A��������������ʣ�HCl��ǿ����ʣ���ͬŨ�ȵĴ����HCl��Һ��pH������>���ᣬ����I�ǵζ���������ߣ�A�����
B��NaOH�ʹ���ǡ�÷�Ӧʱ����ǿ�������Σ���������Һ�ʼ��ԡ�pH=7ʱ����Һ�����ԣ�Ҫʹ��Һ�����ԣ���NaOH�����Ӧ��С��ǡ���������ȫ��ӦʱNaOH����������Եζ��������ĵ�V��NaOH��С��20mL�� B����ȷ��
C��V(NaOH)=10.00mLʱ��������Һ�е�����Ϊ�����ʵ���Ũ�ȵ�CH3COOH��CH3COONa���������̶ȴ��ڴ��������ˮ��̶ȣ���Һ�����ԣ���c��H+��>c��OH-����C(CH3COO-)>C(CH3COOH)����C(CH3COO-)+2C(H+)>C(CH3COOH)+2C(OH-)��C�����
D��V(NaOH)=20.00mLʱ��������Һ�е����ʷֱ��Ǵ����ƺ�NaCl�����������ˮ�⡢�����Ӳ�ˮ�⣬����c��Cl-��>c��CH3COO-����D�����
��ѡB��