ΧβΡΩΡΎ»ί
(11Ζ÷)Ρ≥Μ·―ß–ΓΉι≤…”ΟάύΥΤ÷Τ““Υα““θΞΒΡΉΑ÷Ο(»γΆΦ)Θ§“‘ΜΖΦΚ¥Φ÷Τ±ΗΜΖΦΚœ©ΓΘ
“―÷ΣΘΚ

| | ΟήΕ»(g/cm3) | »έΒψ(Γφ) | Ζ–Βψ(Γφ) | »ήΫβ–‘ |
| ΜΖ“―¥Φ | 0.96 | 25 | 161 | Ρή»ή”ΎΥ° |
| ΜΖ“―œ© | 0.81 | Θ≠103 | 83 | Ρ―»ή”ΎΥ° |
ΫΪ12.5mLΜΖΦΚ¥ΦΦ”»κ ‘ΙήA÷–Θ§‘ΌΦ”»κlmL≈®ΝρΥαΘ§“Γ‘»ΚσΖ≈»κΥι¥…Τ§Θ§ΜΚ¬ΐΦ”»»÷ΝΖ¥”ΠΆξ»ΪΘ§‘Ύ ‘ΙήCΡΎΒΟΒΫΜΖΦΚœ©¥÷ΤΖΓΘ
ΔΌA÷–Υι¥…Τ§ΒΡΉς”Ο « Θ§ΒΦΙήB≥ΐΝΥΒΦΤχΆβΜΙΨΏ”–ΒΡΉς”Ο « ΓΘ
ΔΎ ‘ΙήC÷Ο”Ύ±υΥ°‘Γ÷–ΒΡΡΩΒΡ « ΓΘ
(2)÷Τ±ΗΨΪΤΖ
ΔΌΜΖΦΚœ©¥÷ΤΖ÷–Κ§”–ΜΖΦΚ¥ΦΚΆ…ΌΝΩΥα–‘‘”÷ Β»ΓΘΦ”»κ±ΞΚΆ ≥―ΈΥ°Θ§’ώΒ¥ΓΔΨ≤÷ΟΓΔΖ÷≤ψΘ§ΜΖΦΚœ©‘Ύ ≤ψ(Χν…œΜρœ¬)Θ§Ζ÷“ΚΚσ”Ο (Χν»κ±ύΚ≈)œ¥Β”ΓΘ
aΘ°KMnO4»ή“Κ bΘ°œΓH2SO4 cΘ°Na2CO3»ή“Κ
ΔΎ‘ΌΫΪΜΖΦΚœ©Α¥ΆΦΉΑ÷Ο’τΝσΘ§ά以հ¥” (Χν»κ±ύΚ≈)ΩΎΫχ»κΓΘ’τΝσ ±“ΣΦ”»κ…ζ ·Μ“ΒΡΡΩΒΡ ΓΘ

Δέ ’Φ·≤ζΤΖ ±Θ§ΩΊ÷ΤΒΡΈ¬Ε»”Π‘Ύ Ήσ”“Θ§ Β―ι÷ΤΒΟΒΡΜΖ
ΦΚœ©ΨΪΤΖ÷ ΝΩΒΆ”Ύάμ¬έ≤ζΝΩΘ§Ω…ΡήΒΡ‘≠“ρ « ΓΘ
aΘ°’τΝσ ±¥”70ΓφΩΣ Φ ’Φ·≤ζΤΖ
bΘ°ΜΖΦΚ¥Φ ΒΦ ”ΟΝΩΕύΝΥ
cΘ°÷Τ±Η¥÷ΤΖ ±ΜΖΦΚ¥ΦΥφ≤ζΤΖ“ΜΤπ’τ≥ω
(3)“‘œ¬«χΖ÷ΜΖΦΚœ©ΨΪΤΖΚΆ¥÷ΤΖΒΡΖΫΖ®Θ§ΚœάμΒΡ « ΓΘ
aΘ°”ΟΥα–‘ΗΏΟΧΥαΦΊ»ή“Κ bΘ°”ΟΫπ τΡΤ cΘ°≤βΕ®Ζ–Βψ
Θ®1Θ©ΔΌΖά±©Ζ–Θ®1Ζ÷Θ© άδΡΐΘ®1Ζ÷Θ© ΔΎάδΡΐΜΖΦΚœ©Θ§Ζά÷ΙΤδΜ”ΖΔ2Ζ÷Θ©
Θ®2Θ©ΔΌ…œ≤ψΘ®2Ζ÷Θ© CΘ®2Ζ÷Θ© ΔΎ g Θ®2Ζ÷Θ© Δέ83oC Θ®2Ζ÷Θ© b Θ®2Ζ÷Θ© (3)bcΘ®2Ζ÷Θ©
ΫβΈω

 ‘ΡΕΝΩλ≥ΒœΒΝ–¥πΑΗ
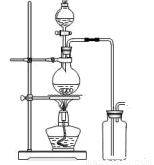
‘ΡΕΝΩλ≥ΒœΒΝ–¥πΑΗ(11Ζ÷Θ©Ρ≥ΩΈΆβ–ΓΉι…ηΦΤΒΡ Β―ι “÷Τ»Γ““Υα““θΞΒΡΉΑ÷Ο»γΆΦ14-2Υυ ΨΘ§A÷–Ζ≈”–≈®ΝρΥαΘ§B÷–Ζ≈”–““¥ΦΓΔΈόΥ°¥ΉΥαΡΤΘ§D÷–Ζ≈”–±ΞΚΆΧΦΥαΡΤ»ή“ΚΓΘ

“―÷ΣΔΌΈόΥ°¬»Μ·ΗΤΩ…”κ““¥Φ–Έ≥…Ρ―»ή”ΎΥ°ΒΡCaCl2ΓΛ6 C2H50H ΔΎ”–ΙΊ”–ΜζΈοΒΡΖ–Βψ:
|
‘ΦΝ |
““Ο― |
““¥Φ |
““Υα |
““Υα““θΞ |
|
Ζ–Βψ/Γφ |
34.7 |
78.5 |
118 |
77.1 |
«κΜΊ¥πΘΚ
(1)≈®ΝρΥαΒΡΉς”Ο « ΘΜ»τ”ΟΆ§ΈΜΥΊ180 ΨΉΌΖ®»ΖΕ®Ζ¥”Π≤ζΈοΥ°Ζ÷Ή”÷–―θ‘≠Ή”ΒΡΧαΙ©’ΏΘ§–¥≥ωΡή±μ Ψ180ΈΜ÷ΟΒΡΜ·―ßΖΫ≥Χ ΫΘΚ ΓΘ
(2)«ρ–ΈΗ…‘οΙήCΒΡΉς”Ο « ΓΘ»τΖ¥”Π«ΑœρD÷–Φ”»κΦΗΒΈΖ”ΧΣΘ§»ή“Κ≥ Κλ…ΪΘ§≤ζ…ζ¥Υœ÷œσΒΡ‘≠“ρ «(”ΟάκΉ”ΖΫ≥Χ Ϋ±μ Ψ) ΘΜΖ¥”ΠΫα χΚσD÷–ΒΡœ÷œσ « Θ°
(3)¥”D÷–Ζ÷άκ≥ωΒΡ““Υα““θΞ÷–≥ΘΚ§”–“ΜΕ®ΝΩΒΡ““¥ΦΓΔ““Ο―ΚΆΥ°Θ§”Πœ»Φ”»κΈόΥ°¬»Μ·ΗΤΘ§Ζ÷άκ≥ω (ΧνΈο÷ Οϊ≥ΤΘ©ΘΜ‘ΌΦ”»κ(¥ΥΩ’¥”œ¬Ν–―Γœν÷–―Γ‘ώΘ§Χν±ύΚ≈) Θ§»ΜΚσΫχ––’τΝσΘ§ ’Φ·77ΓφΉσ”“ΒΡΝσΖ÷Θ§“‘ΒΟΫœ¥ΩΨΜΒΡ““Υα““θΞΓΘ
AΘ°Έε―θΜ·ΕΰΝΉ BΘ°Φν ·Μ“ CΘ°ΈόΥ°ΝρΥαΡΤ DΘ°…ζ ·Μ“
(11Ζ÷Θ©Ρ≥ΩΈΆβ–ΓΉι…ηΦΤΒΡ Β―ι “÷Τ»Γ““Υα““θΞΒΡΉΑ÷Ο»γΆΦ14-2Υυ ΨΘ§A÷–Ζ≈”–≈®ΝρΥαΘ§B÷–Ζ≈”–““¥ΦΓΔΈόΥ°¥ΉΥαΡΤΘ§D÷–Ζ≈”–±ΞΚΆΧΦΥαΡΤ»ή“ΚΓΘ

“―÷ΣΔΌΈόΥ°¬»Μ·ΗΤΩ…”κ““¥Φ–Έ≥…Ρ―»ή”ΎΥ°ΒΡCaCl2ΓΛ6 C2H50H ΔΎ”–ΙΊ”–ΜζΈοΒΡΖ–Βψ:
| ‘ΦΝ | ““Ο― | ““¥Φ | ““Υα | ““Υα““θΞ |
| Ζ–Βψ/Γφ | 34.7 | 78.5 | 118 | 77.1 |
«κΜΊ¥πΘΚ
(1)≈®ΝρΥαΒΡΉς”Ο « ΘΜ»τ”ΟΆ§ΈΜΥΊ180 ΨΉΌΖ®»ΖΕ®Ζ¥”Π≤ζΈοΥ°Ζ÷Ή”÷–―θ‘≠Ή”ΒΡΧαΙ©’ΏΘ§–¥≥ωΡή±μ Ψ180ΈΜ÷ΟΒΡΜ·―ßΖΫ≥Χ ΫΘΚ ΓΘ
(2)«ρ–ΈΗ…‘οΙήCΒΡΉς”Ο « ΓΘ»τΖ¥”Π«ΑœρD÷–Φ”»κΦΗΒΈΖ”ΧΣΘ§»ή“Κ≥ Κλ…ΪΘ§≤ζ…ζ¥Υœ÷œσΒΡ‘≠“ρ «(”ΟάκΉ”ΖΫ≥Χ Ϋ±μ Ψ) ΘΜΖ¥”ΠΫα χΚσD÷–ΒΡœ÷œσ « Θ°
(3)¥”D÷–Ζ÷άκ≥ωΒΡ““Υα““θΞ÷–≥ΘΚ§”–“ΜΕ®ΝΩΒΡ““¥ΦΓΔ““Ο―ΚΆΥ°Θ§”Πœ»Φ”»κΈόΥ°¬»Μ·ΗΤΘ§Ζ÷άκ≥ω (ΧνΈο÷ Οϊ≥ΤΘ©ΘΜ‘ΌΦ”»κ(¥ΥΩ’¥”œ¬Ν–―Γœν÷–―Γ‘ώΘ§Χν±ύΚ≈) Θ§»ΜΚσΫχ––’τΝσΘ§ ’Φ·77ΓφΉσ”“ΒΡΝσΖ÷Θ§“‘ΒΟΫœ¥ΩΨΜΒΡ““Υα““θΞΓΘ
AΘ°Έε―θΜ·ΕΰΝΉ BΘ°Φν ·Μ“ CΘ°ΈόΥ°ΝρΥαΡΤ DΘ°…ζ ·Μ“
(11Ζ÷Θ©Ρ≥ΩΈΆβ–ΓΉι…ηΦΤΒΡ Β―ι “÷Τ»Γ““Υα““θΞΒΡΉΑ÷Ο»γΆΦ14-2Υυ ΨΘ§A÷–Ζ≈”–≈®ΝρΥαΘ§B÷–Ζ≈”–““¥ΦΓΔΈόΥ°¥ΉΥαΡΤΘ§D÷–Ζ≈”–±ΞΚΆΧΦΥαΡΤ»ή“ΚΓΘ
“―÷ΣΔΌΈόΥ°¬»Μ·ΗΤΩ…”κ““¥Φ–Έ≥…Ρ―»ή”ΎΥ°ΒΡCaCl2ΓΛ6 C2H50H ΔΎ”–ΙΊ”–ΜζΈοΒΡΖ–Βψ:
| ‘ΦΝ | ““Ο― | ““¥Φ | ““Υα | ““Υα““θΞ |
| Ζ–Βψ/Γφ | 34.7 | 78.5 | 118 | 77.1 |
«κΜΊ¥πΘΚ
(1)≈®ΝρΥαΒΡΉς”Ο « ΘΜ»τ”ΟΆ§ΈΜΥΊ180 ΨΉΌΖ®»ΖΕ®Ζ¥”Π≤ζΈοΥ°Ζ÷Ή”÷–―θ‘≠Ή”ΒΡΧαΙ©’ΏΘ§–¥≥ωΡή±μ Ψ180ΈΜ÷ΟΒΡΜ·―ßΖΫ≥Χ ΫΘΚ ΓΘ
(2)«ρ–ΈΗ…‘οΙήCΒΡΉς”Ο « ΓΘ»τΖ¥”Π«ΑœρD÷–Φ”»κΦΗΒΈΖ”ΧΣΘ§»ή“Κ≥ Κλ…ΪΘ§≤ζ…ζ¥Υœ÷œσΒΡ‘≠“ρ «(”ΟάκΉ”ΖΫ≥Χ Ϋ±μ Ψ) ΘΜΖ¥”ΠΫα χΚσD÷–ΒΡœ÷œσ « Θ°
(3)¥”D÷–Ζ÷άκ≥ωΒΡ““Υα““θΞ÷–≥ΘΚ§”–“ΜΕ®ΝΩΒΡ““¥ΦΓΔ““Ο―ΚΆΥ°Θ§”Πœ»Φ”»κΈόΥ°¬»Μ·ΗΤΘ§Ζ÷άκ≥ω (ΧνΈο÷ Οϊ≥ΤΘ©ΘΜ‘ΌΦ”»κ(¥ΥΩ’¥”œ¬Ν–―Γœν÷–―Γ‘ώΘ§Χν±ύΚ≈) Θ§»ΜΚσΫχ––’τΝσΘ§ ’Φ·77ΓφΉσ”“ΒΡΝσΖ÷Θ§“‘ΒΟΫœ¥ΩΨΜΒΡ““Υα““θΞΓΘ
AΘ°Έε―θΜ·ΕΰΝΉ BΘ°Φν ·Μ“ CΘ°ΈόΥ°ΝρΥαΡΤ DΘ°…ζ ·Μ“
 H++HCO3-
Ka1 =4Θ°45ΓΝ10-7
H++HCO3-
Ka1 =4Θ°45ΓΝ10-7