��Ŀ����
(11�֣�ij����С����Ƶ�ʵ������ȡ����������װ����ͼ14-2��ʾ��A�з���Ũ���ᣬB�з����Ҵ�����ˮ�����ƣ�D�з��б���̼������Һ��
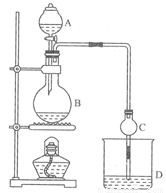
��֪����ˮ�Ȼ��ƿ����Ҵ��γ�������ˮ��CaCl2��6 C2H50H ���й��л���ķе�:
|
�Լ� |
���� |
�Ҵ� |
���� |
�������� |
|
�е�/�� |
34.7 |
78.5 |
118 |
77.1 |
��ش�
(1)Ũ����������� ������ͬλ��180ʾ�ٷ�ȷ����Ӧ����ˮ��������ԭ�ӵ��ṩ�ߣ�д���ܱ�ʾ180λ�õĻ�ѧ����ʽ�� ��
(2)���θ����C�������� ������Ӧǰ��D�м��뼸�η�̪����Һ�ʺ�ɫ�������������ԭ����(�����ӷ���ʽ��ʾ) ����Ӧ������D�е������� ��
(3)��D�з���������������г�����һ�������Ҵ������Ѻ�ˮ��Ӧ�ȼ�����ˮ�Ȼ��ƣ������ (���������ƣ����ټ���(�˿մ�����ѡ����ѡ������) ��Ȼ����������ռ�77�����ҵ���֣��Եýϴ���������������
A������������ B����ʯ�� C����ˮ������ D����ʯ��

��������

 �п�������㾫��ϵ�д�
�п�������㾫��ϵ�д� ������ĩ��ϰ��ѵ��ϵ�д�
������ĩ��ϰ��ѵ��ϵ�д� С��ʿ��ĩ����100��ϵ�д�
С��ʿ��ĩ����100��ϵ�д�(11�֣�ij����С����Ƶ�ʵ������ȡ����������װ����ͼ14-2��ʾ��A�з���Ũ���ᣬB�з����Ҵ�����ˮ�����ƣ�D�з��б���̼������Һ��
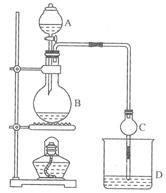
��֪����ˮ�Ȼ��ƿ����Ҵ��γ�������ˮ��CaCl2��6 C2H50H ���й��л���ķе�:
| �Լ� | ���� | �Ҵ� | ���� | �������� |
| �е�/�� | 34.7 | 78.5 | 118 | 77.1 |
��ش�
(1)Ũ����������� ������ͬλ��180ʾ�ٷ�ȷ����Ӧ����ˮ��������ԭ�ӵ��ṩ�ߣ�д���ܱ�ʾ180λ�õĻ�ѧ����ʽ�� ��
(2)���θ����C�������� ������Ӧǰ��D�м��뼸�η�̪����Һ�ʺ�ɫ�������������ԭ����(�����ӷ���ʽ��ʾ) ����Ӧ������D�е������� ��
(3)��D�з���������������г�����һ�������Ҵ������Ѻ�ˮ��Ӧ�ȼ�����ˮ�Ȼ��ƣ������ (���������ƣ����ټ���(�˿մ�����ѡ����ѡ������) ��Ȼ����������ռ�77�����ҵ���֣��Եýϴ���������������
A������������ B����ʯ�� C����ˮ������ D����ʯ��
(11�֣�ij����С����Ƶ�ʵ������ȡ����������װ����ͼ14-2��ʾ��A�з���Ũ���ᣬB�з����Ҵ�����ˮ�����ƣ�D�з��б���̼������Һ��
��֪����ˮ�Ȼ��ƿ����Ҵ��γ�������ˮ��CaCl2��6 C2H50H ���й��л���ķе�:
| �Լ� | ���� | �Ҵ� | ���� | �������� |
| �е�/�� | 34.7 | 78.5 | 118 | 77.1 |
��ش�
(1)Ũ����������� ������ͬλ��180ʾ�ٷ�ȷ����Ӧ����ˮ��������ԭ�ӵ��ṩ�ߣ�д���ܱ�ʾ180λ�õĻ�ѧ����ʽ�� ��
(2)���θ����C�������� ������Ӧǰ��D�м��뼸�η�̪����Һ�ʺ�ɫ�������������ԭ����(�����ӷ���ʽ��ʾ) ����Ӧ������D�е������� ��
(3)��D�з���������������г�����һ�������Ҵ������Ѻ�ˮ��Ӧ�ȼ�����ˮ�Ȼ��ƣ������ (���������ƣ����ټ���(�˿մ�����ѡ����ѡ������) ��Ȼ����������ռ�77�����ҵ���֣��Եýϴ���������������
A������������ B����ʯ�� C����ˮ������ D����ʯ��